In this interesting new article, Markus Hess and Susanne Fleischer describe their technique for managing recurrent laryngeal papillomatosis in an outpatient setting using channelled endoscopes.
The recurrent respiratory papillomatosis (RRP) of the larynx is a chronic HPV-associated viral disease. Clinical management relies on local control with high-resolution distal chip flexible endoscopes, zoom endoscopy techniques like the “dipping“ manoeuvres and spectral image enhancement (e.g. narrow band imaging - NBI).
Biopsies for subsequent histology and viral subtyping can be taken with a channelled flexible endosope in the unsedated patient. Radicality in excision is not feasible due to inherent complications like lifelong vocal fold scarring. Multimodal therapy is offered in an ambulatory setting. Our standard treatment consists of (a) in-office ‘debulking’ with transnasal fiber-guided photoangiolytic laser surgery (445nm wavelength), (b) local administration of antiviral or antiangiogenetic agents (cidofovir or bevacizumab), and (c) therapeutic vaccination. For most of our patients, these in-office procedures are highly appreciated alternatives and preferred to repetitive treatments in general anaesthesia.
“With the video offline review process directly after video recording, we can ascertain – or rule out – the diagnosis of RRP with a high degree of accuracy.”
Diagnosis
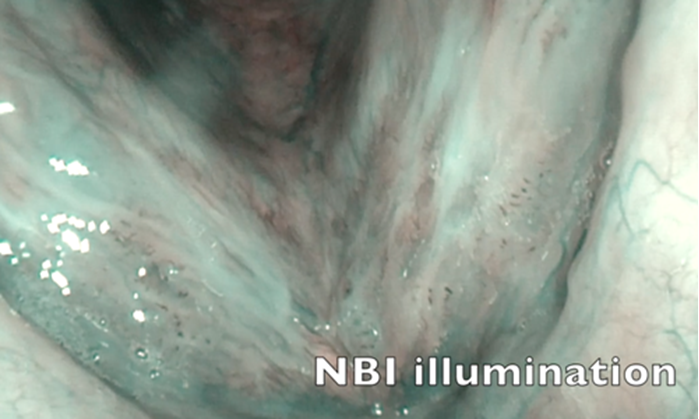
With transnasally-routed high-resolution flexible distal chip endoscopes, we can obtain a close-up view of laryngeal lesions in the awake, topically anaesthetised and unsedated patient. For children, we use a 2mm flexible distal chip endoscope; for adults, a 4.8mm flexible distal chip endoscope. Zoom magnification is achieved with the flexible endoscope forwarded to the glottal level during a prolonged inspiration phase: the dipping maneouvre. This allows for close-to-object endoscopy enabling superb image resolution of tissue surfaces. In combination with additional narrow band imaging (NBI) the diagnostic accuracy can be enhanced during this in-office transnasal, painless endoscopy – even in neonates. Thus, with the video offline review process directly after video recording, we can ascertain – or rule out – the diagnosis of RRP with a high degree of accuracy. In the diagnostic process, we always enforce at least one tissue biopsy for subsequent histological examination, including HPV-DNA subtyping. During repeated office visits, one might reconsider taking follow-up biopsies to make sure there is no malignant transformation.
Therapy
When RRP is diagnosed, it cannot be stated often enough that surgical treatment is not intended as a radical excision technique, since papilloma tend to recur and a radical surgical approach would lead to iatrogenic scarring, which itself can cause (lifelong) hoarseness. Thus, and because RRP surgery is basically mass reduction (debulking), the clinical treatment will benefit from additional measures to eliminate the RRP. Some clinicians think that significant viral load reduction will enable the patient to get to a immunological level of sufficient suppression of local RRP proliferation.
Figure 1. In-office transnasal dipping manoeuvre for high magnification of RRP. NBI is used to better detect RRP vessel patterns.
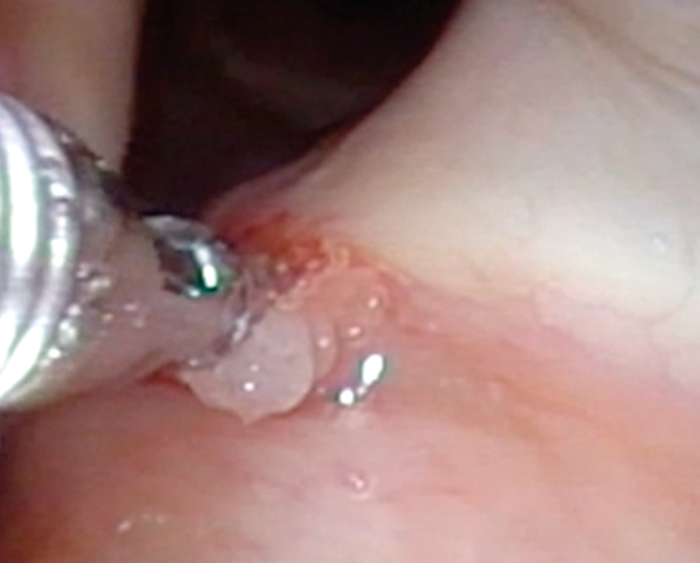
Figure 2. In-office transnasal channelled endoscope. Topical anaesthesia (lidocaine 4%), unsedated patient. Biopsy instrument routed through instrument channel.
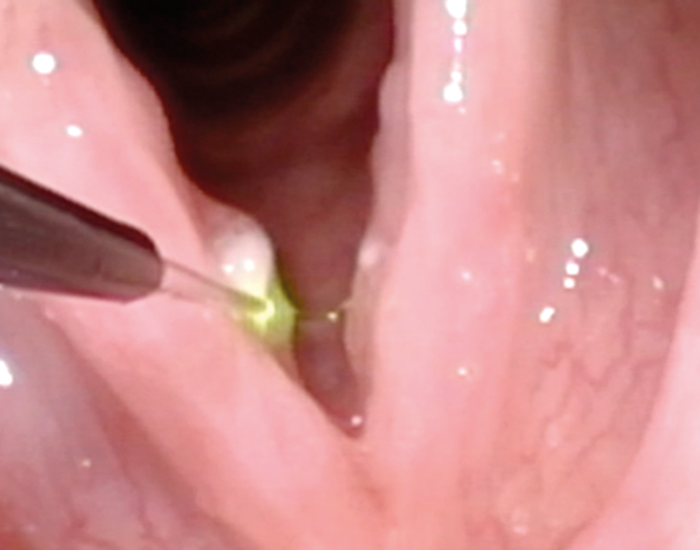
Figure 3. Photoangiolytic laser treatment (445nm blue laser with 400µm glass fibre) routed through instrument channel.
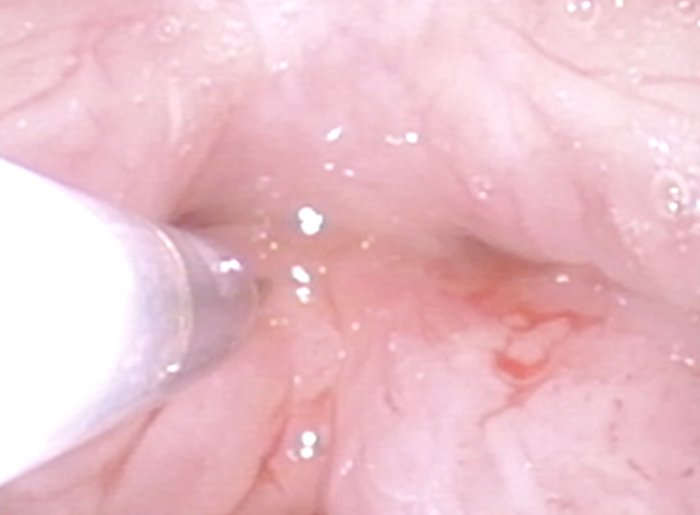
Figure 4. Intralesional injection (here: cidofovir) after laser treatment. Note that endoscope is rotated for easier access to anterior commissure.
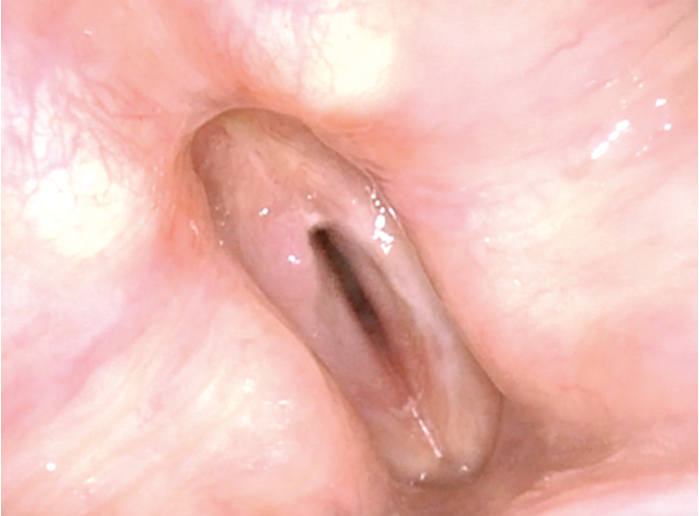
Figure 5. Videostroboscopy assesses post-operative vocal fold pliability.
Laser Treatment
The most important treatment approach for RRP is photoangiolytic laser surgery. The aim of the surgery is reduction of papilloma masses. With photoangiolytic lasers such as KTP (532nm), argon laser (514nm) and blue laser (445nm), only the vessels inside of the papilloma are coagulated (selective photoangiolysis) and this subsequently ‘dries out’ the papilloma within the next hours. Thus, this photoangiolysis is not, per se, debulking the lesion immediately, but it leads to falling off of dead cellular material after subsequent throat clearing, coughing, swallowing and voicing. We now use a 445nm laser (445nm TruBlue®, ARC Laser, Nuremberg, Germany).
With visual monitoring during the lasering and application of higher energy levels, RRP will be blanched (i.e. coagulated) and the blanched material may even be scratched away with the tip of the glass fibre. Higher energy application will lead to carbonisation, which is mostly avoided because of heat sensation which itself might induce gag responses.
When beginning the lasering of RRP, we prefer a ‘staggered approach’ with a slow glass fibre advancement towards the targeted tissue area while pulsing the laser at a frequency of approximately 3-4 pulses per second. At a certain point, the tissue surface will blanch and the treatment starts to be effective. When RRP are located inferior to the vocal folds, inside the Morgagni’s ventricle or at the anterior commissure, we use the ‘rotation laryngoscopy’ manouvre to reach those areas.
This laser treatment can be applied in adults in the office – a painless treatment in an ambulatory surgical setting. In the USA, many patients with RRP receive photoangiolytic laser treatment. However, children mostly do not receive this office-based laser treatment because one cannot expect their adequate cooperation in such a procedure.
Of course, when lasers are used in the office, laser specific safety regulations have to be applied (including appropriate eye protection and special masks).
Adjuvant Treatment
Local injection therapy: After having an informed consent form signed by the patient, we inject antiproliferative agents intralesionally (i.e. in the underlying epithelium) with a tiny cannula routed through the working channel of the flexible endoscope. When using our standard procedure, we recommend intralesional injections of cidofovir (5mg/ml) every six weeks as an off-label pharmaceutical agent. Alternatively, the use of bevacizumab (Avastin®) as an ‘off-label’ antiangiogenetic substance for RRP may be recommended. We have not encountered any side-effects for either substance in more than 30 patients so far.
Vaccination: We recommend the (off-label) therapeutic vaccination with Gardasil® to boost the immunoreactivity.
OUR MULTIMODAL APPROACH
- Diagnostic biopsy for histology and viral subtyping
- In-office fibre-guided photoangiolytic laser therapy
- Cidofovir/bevacizumab injections in six-weeks intervals
- Vaccination according to virus type (mostly Gardasil®)
Conclusion
In-office diagnosis of RRP is facilitated by using distal chip high resolution flexible endoscopy with NBI, in combination with special endoscope handling manoeuvres like rotation laryngoscopy and zoom endoscopy during a ‘dipping’ manoeuvre. Under topical lidocaine spray anaesthesia and with the use of channelled flexible endoscopes, we perform biopsies, fibre-guided photoangiolytic laser surgery and intralesional injections with antiproliferative agents (cidofovir or bevacizumab) during the same intervention in the unsedated patient. We regularly recommend (off-label) adjuvant therapeutic vaccination with Gardasil®. Many patients favour this multimodal in-office approach and prefer this alternative for regularly needed therapeutic interventions to avoid otherwise necessary operations in general anaesthesia.
References
1. Zeitels SM, Akst LM, Burns JA, et al. Office-based 532-nm pulsed KTP laser treatment of glottal papillomatosis and dysplasia. Ann Otol Rhinol Laryngol 2006;115(9):679-85.
2. Mallur PS, Johns MM 3rd, Amin MR, Rosen CA. Proposed classification system for reporting 532-nm pulsed potassium titanyl phosphate laser treatment effects on vocal fold lesions. Laryngoscope 2014;124(5):1170-5.
3. Hess M, Fleischer S. KTP laser in the office. ENT & Audiology News 2016;25(1):106-9.
4. Hess M, Fleischer S, Ernstberger M. New 445 nm blue laser for laryngeal surgery combines photoangiolytic and cutting properties. Eur Arch Otorhinolaryngol 2018;275(6):1557-67.
Declaration of competing interests:
Markus Hess has been consulting with companies (in alphabetical order) as follows: ARC Laser, Laryngograph, Olympus, Pentax, Storz, Wolf, Xion.