Leanne Hamilton and Louise Clark describe their technique for helping to localise parathyoid adenomas. Surgically this can be difficult, so careful preoperative evaluation using imaging as described can help minimise difficulties intraoperatively when identifying the parathyroid adenoma.
Preoperative imaging has historically been critical for parathyroid surgery to answer the following three questions: firstly, to help determine if it is a single gland or multi-gland problem; secondly, to evaluate any concomitant thyroid disease; and lastly to ascertain any ectopic parathyroid gland location. In addition, this imaging can be utilised to pinpoint the parathyroid adenoma, allowing for targeted parathyroid exploration. This article demonstrates a novel method to localise a parathyroid adenoma precisely.
Preoperative scans
As with all parathyroid surgery, ultrasound of neck is used to check thyroid gland integrity. Our preference for location of the parathyroid adenoma is using single-photon emission computed tomography (Spect CT) and nuclear medicine (NM) scans (see Figure 1).
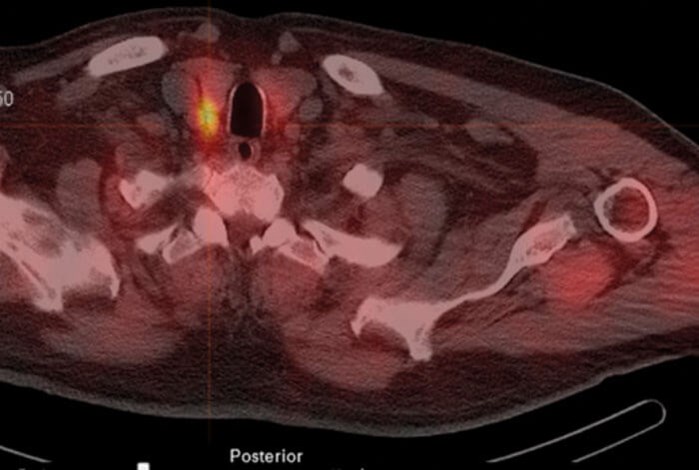
Figure 1. Spect CT scan in axial plane showing position of the parathyroid adenoma highlighted.
Using the PACS software, the corresponding CT images are viewed using multiplanar reconstruction (MPR). The PACS ruler is used to measure the following (see Figures 2-5).
- Size of the parathyroid gland in three dimensions.
- Distance from the cricoid to the parathyroid gland.
- Distance from posterior edge of thyroid to the parathyroid gland.
- Distance from the skin to the parathyroid gland.
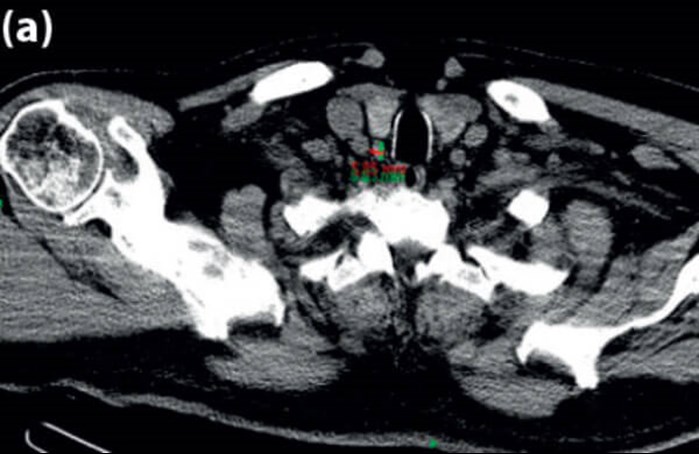
(a).
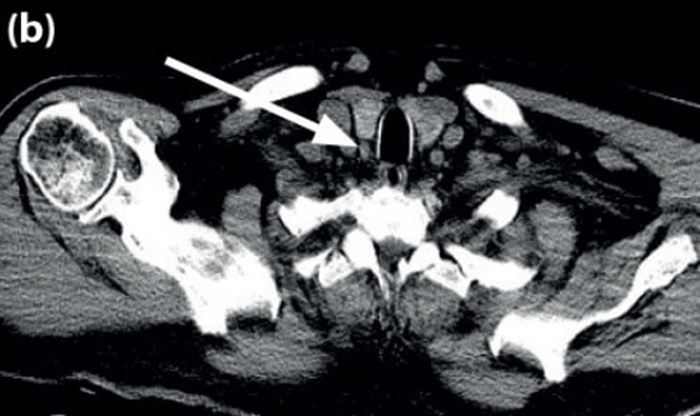
(b).
Figure 2 (a) Axial CT scan showing PT gland measurements 5.85mm x 8.83mm; (b) arrow points to PT gland.
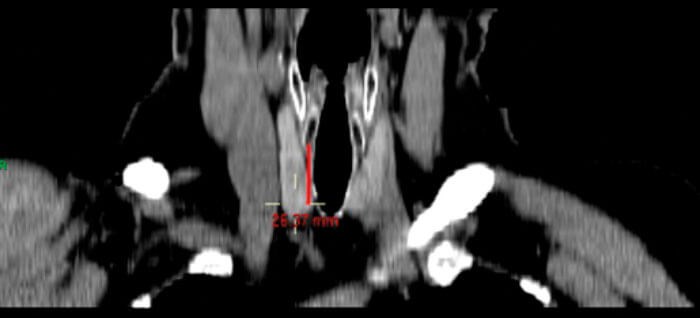
Figure 3. Coronal CT image on MPR setting showing distance of PT gland from cricoid (26.37mm).
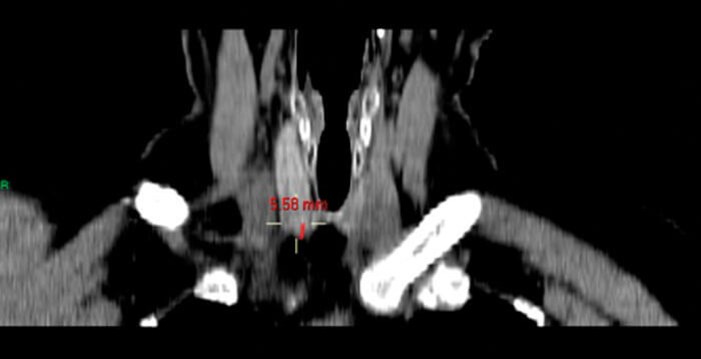
Figure 4. Coronal CT image to show measurement from posterior inferior edge of thyroid gland to PT gland (5.58mm).
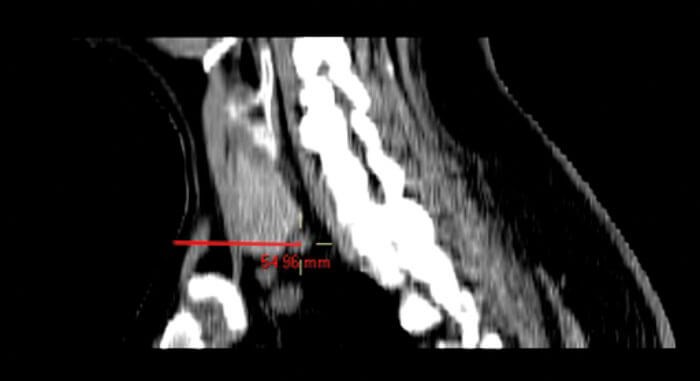
Figure 5. Sagittal CT image showing distance from skin to PT gland (54.96mm), also confirms distance between inferior edge of thyroid gland to PT gland.
Operative procedure (see Figures 6-11)
Consent for the procedure will take into account risks of surgery including scar, bleeding, infection, damage to recurrent laryngeal nerve (RLN), failure of procedure, and postoperative hypocalcaemia versus the option of not doing surgery and the consequential effects of ongoing hypercalcemia on the body. The patient is positioned with neck extended and a NIM monitor used for RLN monitoring. No local anaesthetic infiltration is used prior to incision. Intraoperative parathyroid hormone levels are not measured routinely as it adds to the cost and duration of the procedure and is usually not required with good preoperative imaging. The preoperative measurements are used intraoperatively. At the start of the procedure, a ruler and marker pen is used to measure the distance from cricoid, and this helps guide incision level on the neck.
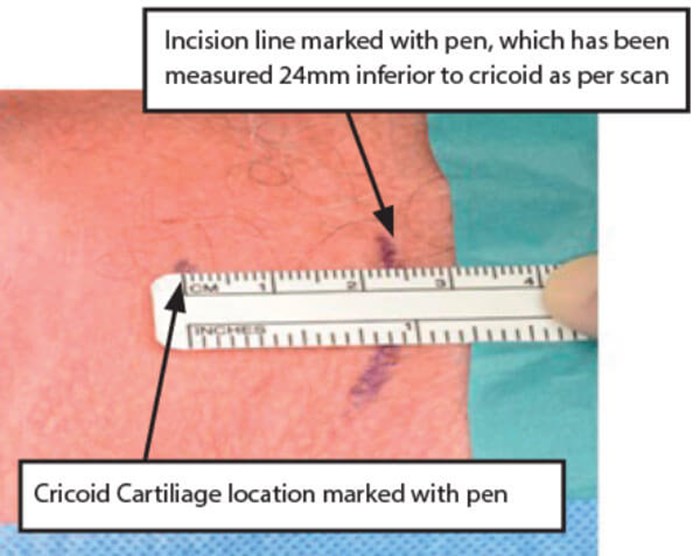
Figure 6. Ruler used to measure ‘PT gland to Cricoid’ distance for skin incision.
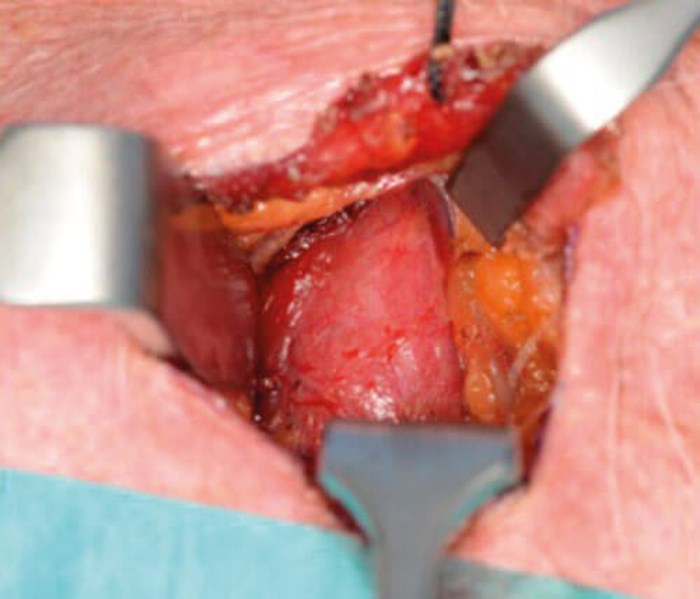
Figure 7. Identification of thyroid gland surface.
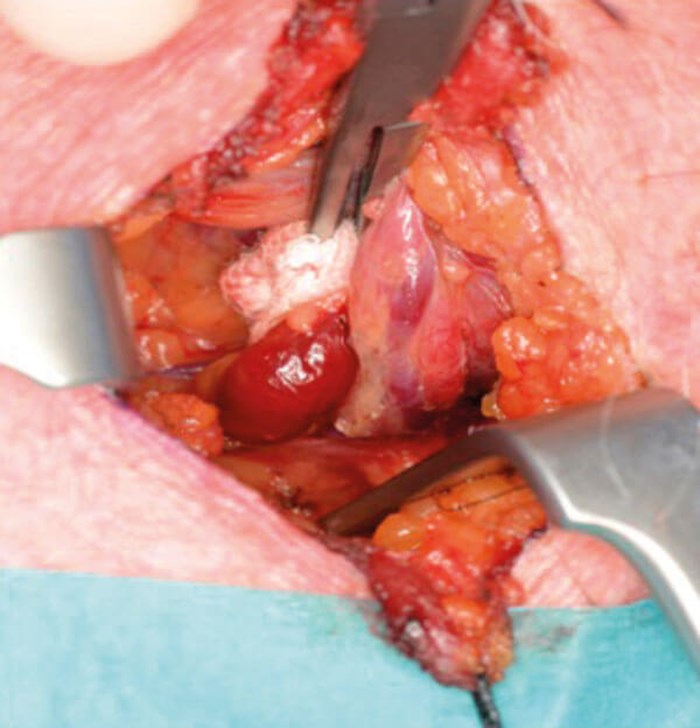
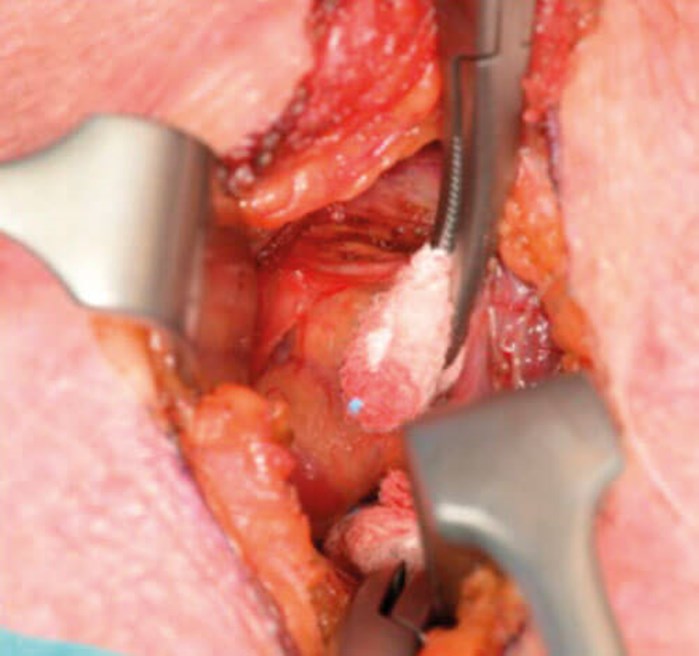
Figure 8 and 9. Minimal dissection required. Retraction of thyroid lobe to locate PT gland.
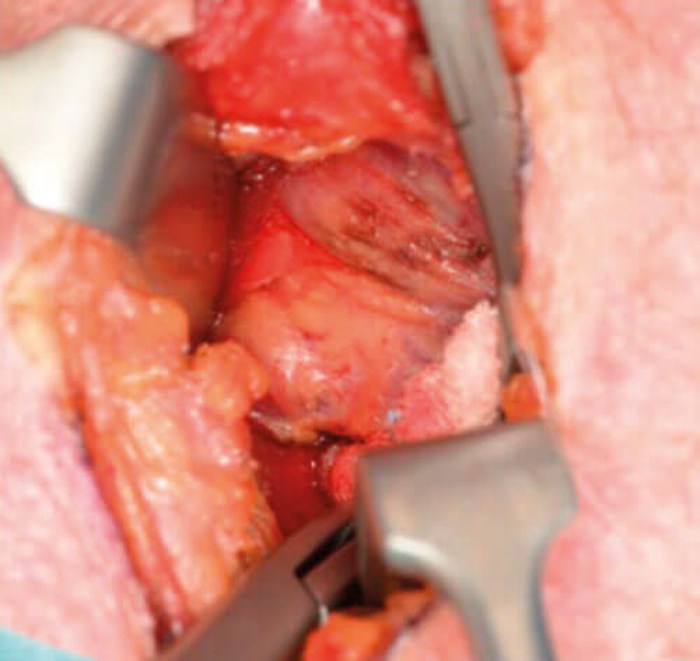
Figure 10. Identification of the PT gland in the exact predicted location.
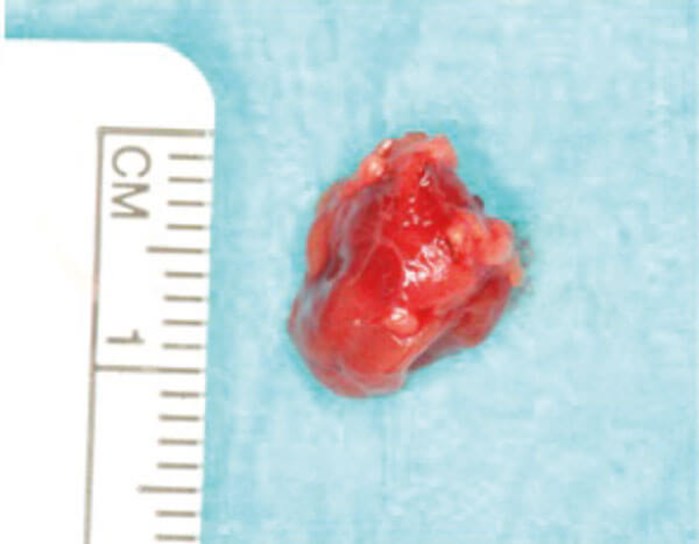
Figure 11. The PT gland specimen.
“The primary advantage of this method of targeted location is the reduced risk to surrounding structures as the dissection area is minimised”
When the dissection has progressed to the stage of identifying the surface of the thyroid gland, the ruler can be used to measure the distance from posterior aspect of thyroid to where the targeted PT gland should be. Confirmation of cricoid distance may be used at this time to accurately pinpoint the adenoma. This is useful when starting to do this type of surgery.
Discussion
There are several advantages to using this technique. The primary advantage of this method of targeted location is the reduced risk to surrounding structures as the dissection area is minimised. Therefore, there is a reduced risk of damage to the recurrent laryngeal nerve and to healthy PT glands. Targeted location makes the procedure more efficient and reduces the risk of failure of operation as it will be unlikely to remove the wrong gland given the correct measurements from the scan.
Limitations to this method may be encountered in multi-gland disease, albeit this only accounts for 10% cases in the literature. However, the method is still useful even in these cases as it gives accurate measurements for both PT glands and a logical plan for dissection if glands are visible on scans that do not light up on SPECT. The authors had a case which demonstrated this. A 38-year-old lady with primary hyperparathyroidism had bilateral parathyroid masses identified on preoperative scans (SPECT CT and sestimibi).
The mass on the right was intrathyroidal and was a hotspot on the scan, whereas the one on the left was inferior to thyroid and not a hot spot. The surgeon decided to take a baseline PTH and start with targeted removal of the right mass as guided by the measurements. The intraoperative PTH only reduced by 50% at 55 mins intraop, so a decision was made to do a focused exploration on left, again using the measurements to locate the mass. Repeat intraoperative PTH was reduced tenfold at 110min.
“Limitations to this method may be encountered in multi-gland disease, albeit this only accounts for 10% cases in the literature”
Histopathological results from this case confirmed a hypercellular parathyroid gland on the right with thyroiditis, and the mass on the left was a parathyroid carcinoma. This case reminds us that thyroiditis may have an effect on the metabolic imaging, and all masses may potentially need explored. The principles of measurements using preoperative scans for targeted parathyroidectomy were still important in this case of bilateral disease with background thyroiditis as it simplified the dissection required in an otherwise difficult surgical field.
The quality of the radiological images are important to allow accurate measurement and the author acknowledges that this may be a possible limitation in some centres depending on resources. In our centre, previous review of radiological studies done by the author show a sensitivity rate of 92% for SPECT CT and 97% for sestamibi.
Indications for parathyroid surgery for primary hyperparathyroidism are well-documented in guidelines. Surgery for subclinical parathyroid adenoma where the patient is asymptomatic is less known but has been advised to reduce the morbidity of associated outcomes of hypercalcaemia, such as osteoporosis and atherosclerosis. There is growing evidence for reversible aspects of primary hyperparathyroidism with surgical management.
The Montgomery ruling for consent has made this an issue to consider. If it is known that a patient has asymptomatic hyperparathyroidism, they should thus be informed of the potential damage it is already doing to their skeletal, renal and cardiovascular systems before end-stage disease is apparent. The patient must be given the opportunity to make an informed decision regarding medical management or the option of surgery for treatment but can be more confident in the recommendation for early surgery with experienced surgeons. Having a targeted approach may offer an efficient, safer and less invasive surgery.
FURTHER READING
1. Smith D. Parathyroidectomy. ENT & Audiology News 2017;26(3):104-6.
2. Bilezikian JP, Brandi ML, Eastell RJ, et al. Guidelines for the management of asymptomatic primary hyperparathyroidism: summary statement from the Fourth International Workshop. Clin Endocrinol Metab 2014;99(10):3561-9.
Declaration of Competing Interests: None declared.






