Jaclyn Tan interviews members of the London-based multidisciplinary hearing team, about their insights to auditory neuropathy spectrum disorder, and their vision for the future management of this challenging range of conditions.
Auditory neuropathy spectrum disorder (ANSD) describes a wide range of conditions causing varying severities of hearing loss. It accounts for 10% of children with permanent hearing loss. It is difficult to detect ANSD via national hearing screening programmes, and outcomes of current treatments vary. Some forms of ANSD have a genetic cause and, in this field, rapid progress is being made in developing gene therapies to correct gene mutations. We hear from a team of clinicians who care for families with children with ANSD. They have initiated a project to raise awareness and educate clinicians and families about the clinical characteristics of ANSD, the importance of genetic testing, and the opportunities that inner ear gene therapies may offer in the future.

Chrysa Spyridakou, Consultant Audiovestibular Physician
What is ANSD?
The term ‘auditory neuropathy spectrum disorder’ (ANSD) [1] was introduced for patients with hearing problems who have a combination of recordable otoacoustic emissions or cochlear microphonics and absent or atypical auditory brain stem responses. Their behavioural hearing assessments vary from mild to profound hearing loss, and their comprehension of speech is typically worse than predicted. Their outcomes of treatment are also hard to predict based on audiological assessments.
Could you describe the current UK newborn hearing screening programme (NHSP) and how it may fail to detect ANSD?
The UK NHSP pathway is well established (Figure 1), but by using automated otoacoustic emissions (AOAE), well babies with clear responses will not proceed to have an auditory brainstem response (ABR) test. However, they still may have ANSD, and this can only be picked up via an ABR test.
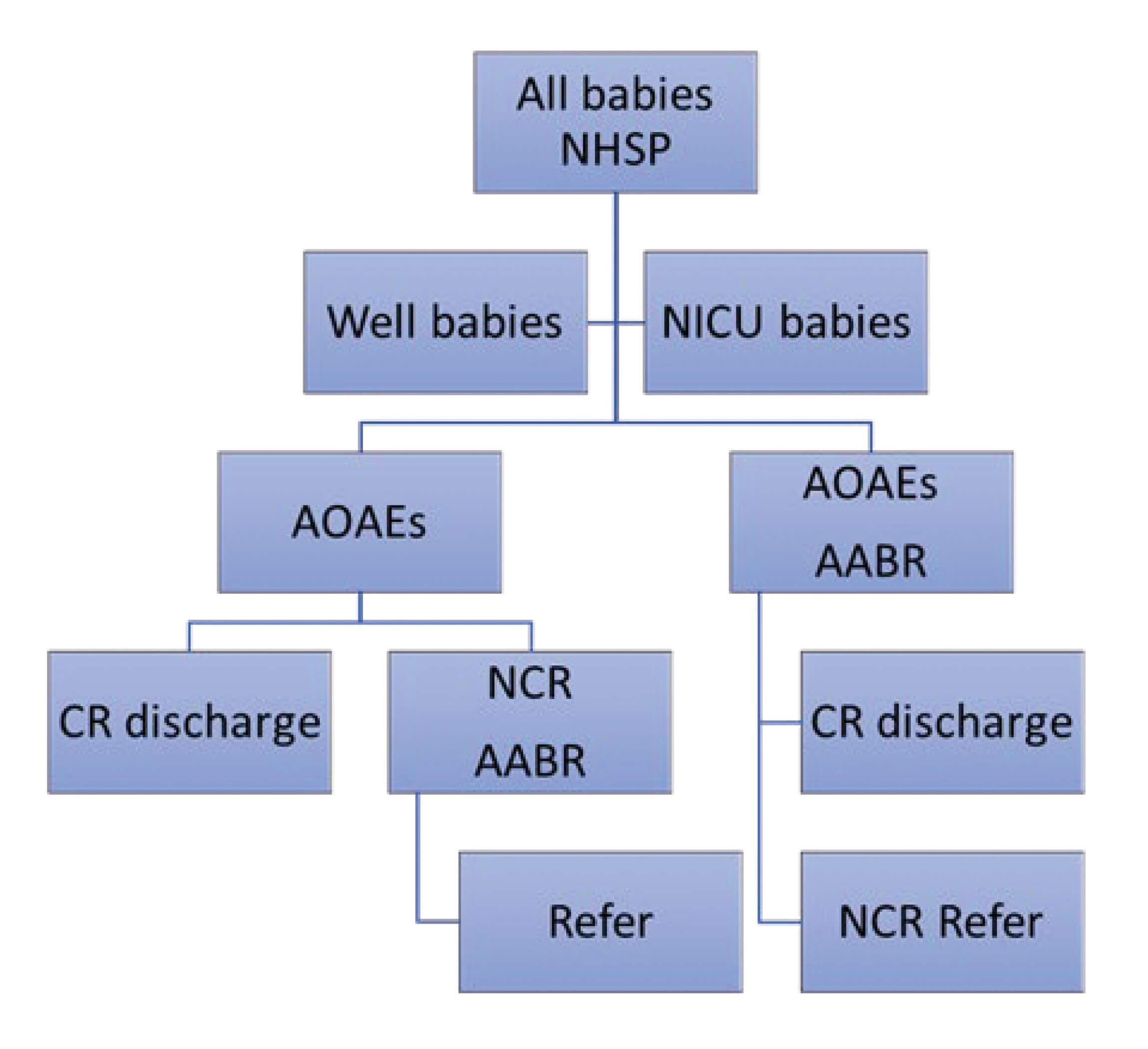
NHSP (newborn hearing screening programme);
AOAE (automated otoacoustic emissions test);
AABR (automated auditory brainstem response);
CR (clear response);
NCR (no clear response).
Figure 1. Current UK NHSP pathway for well babies and neonatal ICU (NICU) babies.
What is your role as an audiovestibular physician for patients with ANSD?
My role is to investigate the causes of ANSD, manage the child holistically and monitor how ANSD impacts a child’s development. Since ANSD is a complex diagnosis, I rely on clinical history and examination before requesting further investigations. All children with ANSD will have an MRI scan of the inner ears and brain.
Based on the current hearing screening pathway, I mainly see referrals from neonatal intensive care (NICU) who routinely have an ABR. In NICU babies with abnormal ABRs but present OAEs, ANSD was previously assumed to be acquired due to prematurity, hypoxia or jaundice. However, we have recently been able to build a much better understanding of the genetic causes of ANSD. Now, in all referred babies including those from NICU, we request gene panels to see if there is an underlying genetic cause.

Robert Nash, Consultant Otologist
Could you describe your role as an otologist within the team treating children with ANSD?
I see children who have hearing loss for a variety of reasons. A small number of children whose hearing is severely affected by ANSD will need cochlear implantation. These children with hearing loss and other ear issues are referred to our cochlear implant centre by audiovestibular physicians, ENT surgeons and audiology departments. They come to us at various ages. Generally, the sooner a child obtains the right treatment, the better its outcome.
What are the outcomes of cochlear implants for children with ANSD?
Cochlear implants rely on the cochlear nerve to transmit hearing to the brain - ANSD describes conditions that cause problems with this nerve. For children with ANSD who require cochlear implantation, outcomes are generally poorer than for those who have other causes of hearing loss.
Any thoughts on gene therapies?
Whilst hearing aids and cochlear implants have changed the lives of millions of people for the better, they are not perfect. It will be fascinating to see how effective hearing loss gene therapies will be - will they restore hearing function without the need to wear devices? If so, they may have the potential to revolutionise the treatment of hearing loss in the same way that cochlear implants did around 30 years ago.

Emma Clement, Consultant in Clinical Genetics and Genomic Medicine
What is your role as a clinical geneticist for patients with ANSD?
In our otogenetics team, I work with audiovestibular physicians and ENT surgeons to review patients affected by ANSD and other forms of hearing loss to consider whether an underlying genetic diagnosis is likely. We may arrange genetic testing or help with the interpretation of these tests. A genetic diagnosis can help families in several ways: it informs risk in future children, treatment decisions, and eligibility for upcoming trials of gene therapy.
What is inner ear gene therapy and how does it work?
You need to address the underlying genetic cause to cure genetic forms of hearing loss. This can be done by providing a genetic substitute for a non-functioning gene (introducing a transgene) (Figure 2). Typically, this would use a viral vector to exploit the host cell replication machinery to copy the introduced genetic material. Other strategies include using substances such as antisense oligonucleotides (ASOs) to target the faulty product of a defective gene, or targeted gene editing using CRISPR-Cas9 to correct the specific genetic fault.
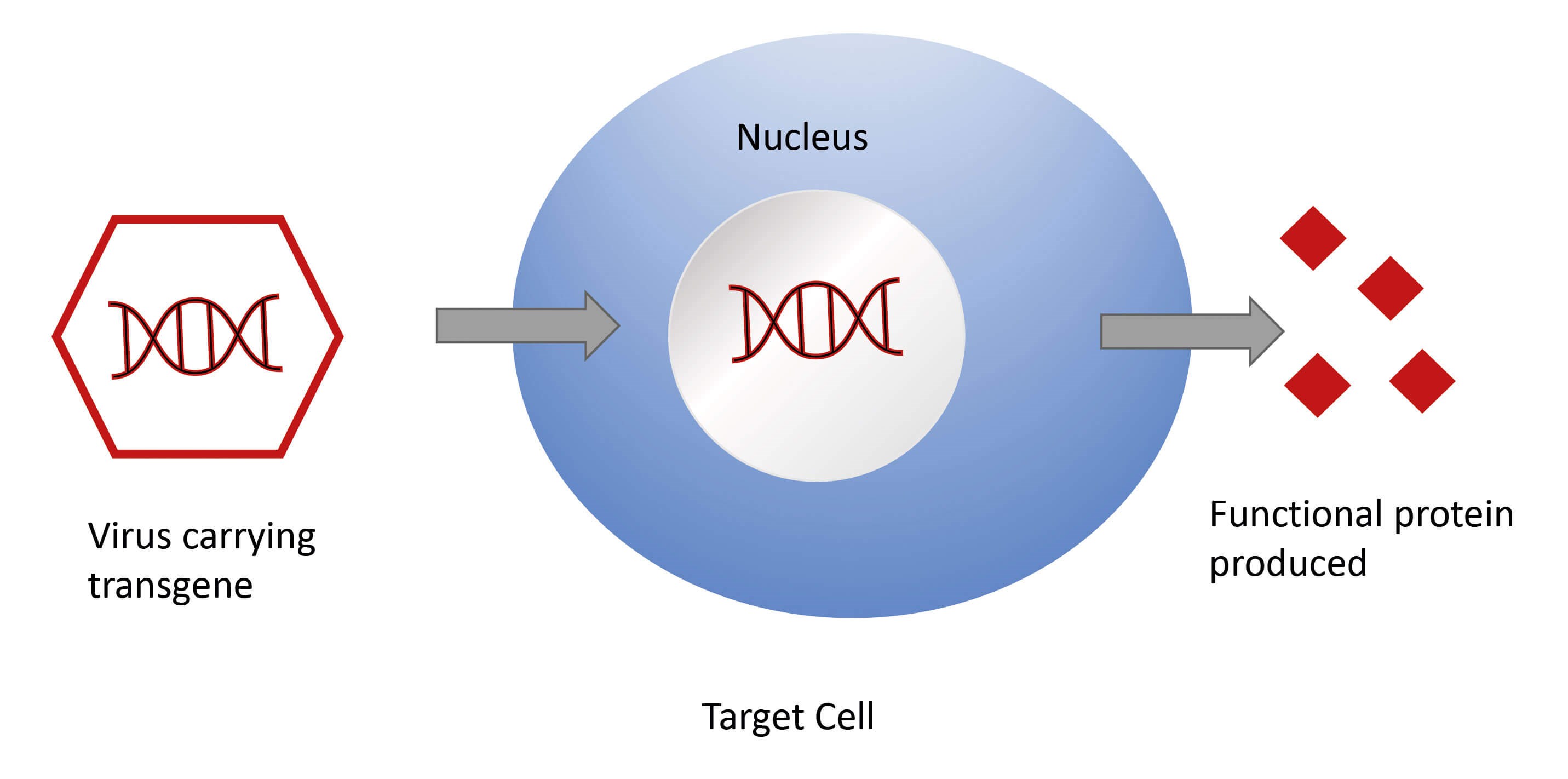
Figure 2. Viral vectors in gene therapy.
Who may benefit from gene therapy and what is the optimal time to do this?
There is ongoing research in developing gene therapy for several genetic forms of hearing loss, including otoferlin (OTOF) mutations, the most common genetic cause of ANSD. OTOF is a good candidate for gene therapy because the sensory epithelium develops normally. The hearing impairment is due to failure of sound-evoked neurotransmitter release with subsequent degeneration of outer hair cells. This is important as it means that there is likely to be a window of opportunity for successful gene therapy before significant cellular degeneration occurs. In cases of congenital hearing loss, timely therapy will also enable children to benefit developmentally from early access to speech and language.
Will inner ear gene therapy be incorporated into practice down the road?
Studies on mice have shown promising results for some forms of genetic hearing loss including OTOF-related ANSD. It will take time before gene therapy can become a treatment option for ANSD. First, we will have to wait for the outcome of the trials.


Nish Mehta, Consultant ENT & Auditory Implant Surgeon
Anne GM Schilder, Professor of Otorhinolaryngology
How is the research and clinical work on genetic hearing loss linked to the National Institute for Health Research Biomedical Research Centre (BRC) at UCLH?
Research on the genetic causes of hearing loss and gene therapies is at the heart of our BRC Hearing Health Theme. Our programme brings together the UCL Ear Institute, Royal National ENT Hospital and Great Ormond Street Hospital to develop, test and deliver innovative treatments for hearing loss. This ranges from deep geno-phenotyping hearing loss and discovery of therapeutic targets to clinical trials and implementation of science to prepare hearing services for these exciting new treatments. We are very fortunate to work in an environment that is at the forefront of genetics and gene therapy. UCLH has recently been awarded a major research grant to establish a centre for advanced therapies for neurological and inner ear disorders. This centre will allow us to expand our research and translate its findings into new treatments for patients.
Reference
1.Hayes D, Sininger Y. Guidelines: Identification and management of infants and children with auditory neuropathy spectrum disorder. Guidelines Development Conference at NHS 2008, Como, Italy 2008:3-8.
www.childrenscolorado.org/
globalassets/departments/ear-nose
-throat/ansd-monograph.pdf
Last accessed August 2022.
Acknowledgement: We would like to acknowledge funding from NIHR UCLH BRC Deafness and Hearing Theme.










