We all have patients who give us a ‘heartsink’ feeling; for many of us, the coughing patient is exactly such a situation. In this overview, Bhaskar Ram and Sangeeta Maini outline their approach to diagnosis.
Declan Costello, Editor.
Introduction
Chronic cough can result in significant morbidity and affects quality of life. The treatment of chronic cough often requires multidisciplinary input. Of increasing interest is the role of cough receptors in cough pathogenesis which will be discussed in the review.
Definition
Chronic cough is defined as lasting eight weeks or longer in adults. Cough lasting less than three weeks is defined as acute and 3-8 weeks sub-acute. The acute and sub-acute cough are often self resolving and mainly post-infective and will not be further discussed in the review.
Cough can be classified by its duration, character, quality, and timing. However, the most useful classification for clinical diagnosis and management purposes is the duration of cough.
Mechanism
Cough is a protective automatic arc reflex and has three components: an afferent sensory limb, a central processing centre, and an efferent limb.
The trigeminal, glossopharyngeal and vagus nerves supply the afferent pathways for cough receptors; the vagus through its pharyngeal, superior laryngeal and pulmonary branches supplies the large majority of these receptors. Receptors are located throughout the airway from the pharynx to the terminal bronchioles, with the greatest concentration located in the larynx, carina and the bifurcation of larger bronchi.
Three types of receptor are predominant [1-3]:
- Rapidly adapting receptors (RARs) that respond to mechanical stimuli
- Slowly adapting receptors (SARs)
- C-fibres that respond to chemical stimuli.
Afferent impulses are transmitted to the cough centre of the brain, located in the medulla of the brainstem, which is connected to the central pathways. Efferent impulses leave the medulla and travel via the vagus, phrenic and spinal motor nerves of C3 to S2.
The cough reflex has been shown to have neuroplasticity such that a hyper-sensitive response is elicited over time due to the cough itself inducing chronic irritation and inflammation and tissue remodelling [4]. Both peripheral and central sensitisation can account for an exaggerated cough response that is common in patients and further contributes to the maintenance of chronic cough [3].
Recently, the term chronic cough hypersensitivity syndrome has been proposed as the new way to label chronic cough, owing to the fact that the underlying abnormality leading to chronic coughing is an abnormally sensitive cough reflex [4-6].
Morice (2013) has suggested a different paradigm in which cough is viewed as the primary condition characterised by afferent neuronal hypersensitivity and different aspects of this syndrome are manifest in the different phenotypes of cough [7].
Aetiology
Prospective studies have shown that three conditions account for the etiologic cause of chronic cough in up to 95% of immunocompetent nonsmoking patients with normal chest x-rays [8]. In order of frequency, they are as follows:
1. Upper airway cough syndrome (UACS), previously referred to as postnasal drip syndrome (PNDS)
2. Asthma
3. Gastroesophageal reflux disease (GERD).
These three conditions make up what is called the pathogenic triad of chronic cough.
Upper airway cough syndrome or post nasal drip syndrome
Postnasal drip syndrome is defined as sensation of secretions from the nose and sinuses that drain into the pharynx with frequent throat clearing. Clinical examination including nasal endoscopy is often normal and non-specific. Occasionally, thick mucoid secretions draining from the posterior nasal cavity into the pharynx with a ‘nodular’ or ‘cobble stone’ appearance of the nasopharynx may be seen.
The reported incidence of rhinitis and postnasal drip vary enormously between different cough clinics 8-58%. Although postnasal drip has a high sensitivity index of 100%, the specificity is often quite low, 37-68%.
This is probably for two reasons. Firstly, the difficulty in defining and therefore quantifying post nasal drip syndrome and, secondly due to a difference in the patient reporting and understanding of symptoms.
It seems likely that the upper airway inflammation reflects inflammation in the area of the larynx which in turn stimulates the cough receptors in the larynx resulting in exacerbation of cough rather than there being any physical ‘dripping’ onto the larynx. Hence, postnasal drip syndrome, is now referred to as ‘upper airway cough syndrome’ (UACS).
Aetiology of upper airway cough syndromes (UACS) includes rhinosinusitis, allergic rhinitis and vasomotor rhinitis.
Chronic rhinosinusitis
Patients with chronic rhinosinusitis often present with nasal congestion, postnasal discharge and rarely with facial pain or chronic cough. Nasal endoscopy may show polypoidal edematous changes in the middle meatus with or without mucopurulent discharge draining into the nasopharynx. Treatments commonly include topical steroid sprays / drops and / or long-term oral antibiotics. Endoscopic sinus surgery is reserved for patients with failed medical therapy [9].
Allergic rhinitis
Patients with allergic rhinitis / upper airway nasal symptoms should be given a therapeutic trial of intranasal steroid and / or antihistamines. Whilst there is little clinical trial evidence available, there is anecdotal evidence that the older agents have greater efficacy than some of the more modern non sedating antihistamines. One possible explanation for this observation is that many of the modern highly specific H1 antagonists have been designed and shown to block only the H1 receptor. Recent research has shown that there are at least four histamine receptors with the H3 receptor playing a possible important role in allergic inflammation.
Vasomotor rhinitis
Vasomotor rhinitis is a common condition seen in the elderly population who present with severe postnasal drip and chronic cough, often affecting the quality of life. It is believed to be due to an imbalance in the autonomic system resulting in parasympathetic hyperactivity. Patients often complain of severe watery rhinorroea following exposure to perfumes, change in weather, alcohol ingestion etc.
Treatment includes topical ipratropium bromide spray or possibly bilateral endoscopic videan neurectomy in resistant cases [10].
Reflux related cough
GERD is implicated in up to 41% patients with chronic cough [11]. The following two mechanisms have been postulated for GERD-associated cough [12].
- Distal esophageal acid exposure that stimulates an esophageal-tracheobronchial cough reflex via the vagus nerve
Microaspiration of esophageal contents into the laryngopharynx and tracheobronchial tree.
The second entity refers to laryngopharyngeal reflux (LPR) or extraesophageal GERD, and it differs from traditional GERD in that it does not manifest as heartburn and tends to occur when the patient is upright as opposed to lying flat. This silent GERD can be present in as many as 75% of patients with chronic cough [6]. Symptoms of LPR include throat clearing, hoarseness and globus sensation. Empiric treatment includes acid suppression and lifestyle and dietary modifications.
The criterion standard for diagnosis of GERD is dual-channel 24-hour pH probe monitoring. Alternatively, flexible naso-pharyngoscopy can reveal glottic changes associated with reflux. These changes are non-specific and often non-diagnostic and include laryngeal edema and erythema, laryngeal pseudosulcus and posterior commissure hypertrophy or pachydermia.
Simply because of the percentages, empiric therapy with acid suppression and lifestyle and dietary modification has been advocated as initial management instead of testing, which is reserved for refractory cases. Lifestyle modifications include limiting fat intake, avoiding caffeine, chocolate, mints, citrus products, alcohol and smoking; and limiting exercise / postures that increase intra-abdominal pressure [13].
The choice of acid suppressive medication can include histamine 2 (H2) blockers, proton pump inhibitors (PPIs), and prokinetic agents. However recent guidelines recommend maximal medical therapy as twice daily PPI with concurrent lifestyle and dietary modification [13]. Although response can be seen in as little as two weeks, at least a 6-8 week trial is needed to fully evaluate a response to treatment, with some patients requiring as long as six months [14, 15].
Asthma syndromes
Asthma and their variants account for 25% of chronic cough [16].
Classic asthma
In classic asthma there is evidence of variable airflow obstruction. Such evidence is easy to obtain in ordinary clinical practice by asking patients to fill out a home diary record card with peak flow monitoring, readings being made morning and evening. Diurnal variation revealed as a sort of pattern on the peak flow chart is characteristic of classic asthma. Morning dipping first described by Sir John Floyer 400 years ago is a cardinal feature.
However, such obvious diagnostic clues are rare in patients with isolated asthmatic cough. A much more frequent pattern is variability in peak flow of a small degree (less than 10%) without particular pattern. On starting anti-asthma medication variability decreases and often there is a gradual rise in peak flow compared with pre-treatment baseline. Final peak flow pattern is one without variability. Because of the difficulty in definition of ‘asthmatic cough’ the term cough preponderant asthma has become established and the asthmatic syndromes without bronchoconstriction are discussed below.
Cough preponderant asthma
In 1986 Irwin described six patients with cough who responded to anti-asthma treatment but who did not have bronchoconstriction. When tested for bronchial hyper-responsiveness these patients exhibited typical asthmatic responses. Thus, he defined what he called cough variant asthma as cough responding to anti-asthma treatment in the presence of bronchial hyper-responsiveness but without bronchospasm. This entity is commonly seen in the cough clinic. Because there is a lack of significant bronchoconstriction many referring clinicians put off the diagnosis of asthma. Clues that this is the correct diagnosis may be found in the history. There is often a history of atopy and either childhood eczema or hay fever. This may be present in either the patient or a first degree relative.
Key to the diagnosis of cough preponderant asthma is the response to therapy. Corticosteroids are the cornerstone of this therapeutic trial. Inhaled corticosteroids usually provide at least some clinical response. In the absence of any other obvious diagnosis then it is permissible to undertake a therapeutic trial of oral prednisolone, 20mg a day for two weeks. The patient should be fully informed that this is a therapeutic trial to see whether there is any reduction in cough. There is a small but significant minority of patients with cough preponderant asthma who only respond to parenteral treatment with steroids. If such a response does occur then attempts to wean oral steroids using second line agents such as leukotriene antagonist, long acting beta agonist are appropriate.
Eosinophilic bronchitis
The term eosinophilic bronchitis is reserved for patients who again respond to anti-asthma medication but do not exhibit either bronchoconstriction or bronchial hyper-responsiveness. As the term implies, sputum examination reveals predominant eosinophils. Patients with eosinophilic bronchitis may be relatively resistant to anti-asthma therapy, only responding to high doses of oral steroids. Attempting to control the disease is important since a proportion of these patients do progress to develop fixed airflow obstruction or bronchiectasis.
Rare causes
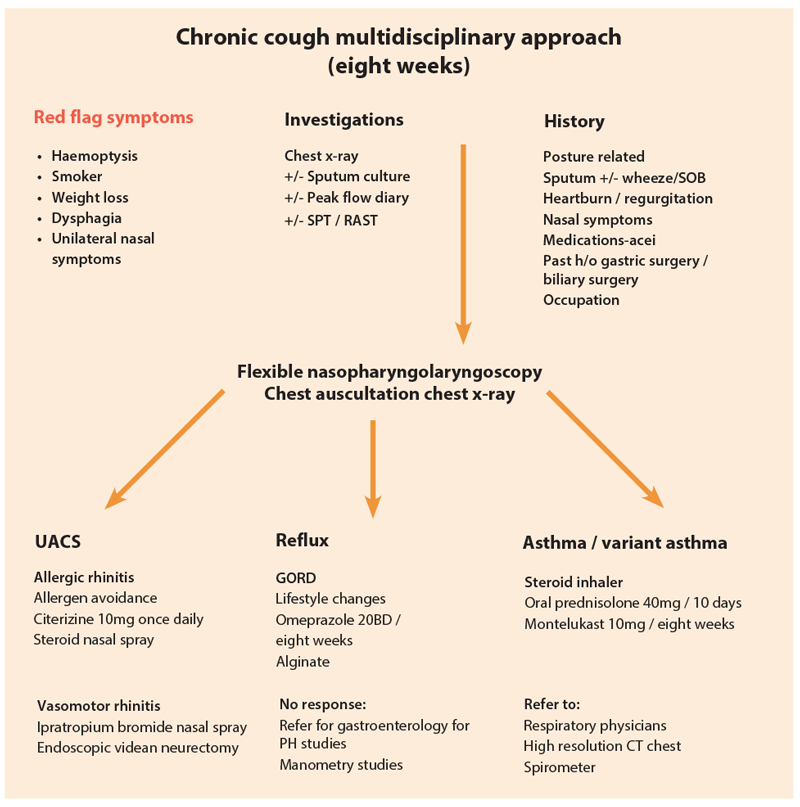
Apart from these three main causes, one needs to consider other rare chest conditions including lung cancer, COPD, bronchiectasis. Lung cancer accounts for 0.2% of chronic cough. We recommend that all patients with chronic cough must have a chest x-ray. Any patient with red flag symptoms such as haemoptysis, unilateral neck mass, unexplained weight loss, breathlessness, chest pain should be immediately referred to his / her respective specialty for further investigations and treatment.
Most causes of cough can be divided into one of the following five categories:
1. Environment irritants and drugs (such as ACE inhibitors)
2. Aero-digestive tract pathology
3. Psychogenic
4. Systemic – rare
5. Idiopathic – rare.
Systematic approach
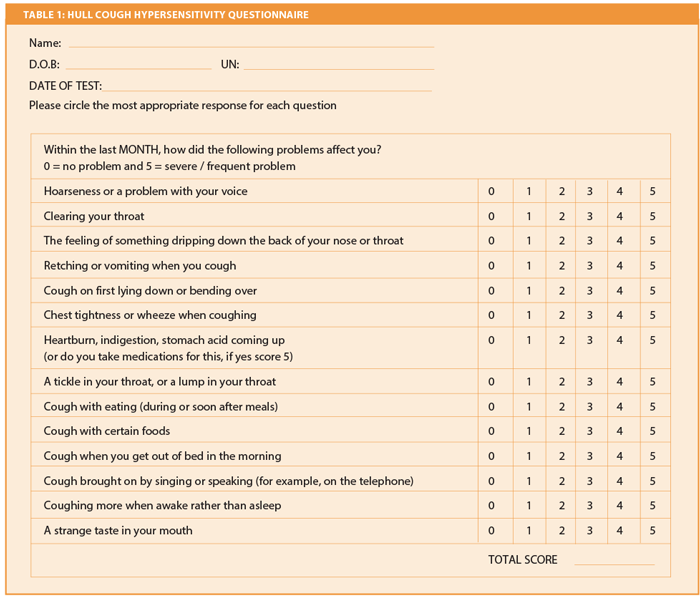
In clinical practice we have found the Hull Cough questionnaire to be very useful in identifying the cause of chronic cough (Table 1). The questionnaire is simple to fill in, is validated and includes 14 key questions related to chronic cough. Symptoms are rated on a scale from 0-5. The maximum score is 70. Normal people score an average 4/70 and the upper limit of normal is 13/70. Patients with a higher score of more than 13 are likely to have ‘cough hypersensitivity syndrome’. Other relevant questions that must be elicited in the history include any smoking, use of ACE inhibitors, occupational history and previous gastric or biliary surgery.
All patients should undergo a chest examination, ENT examination, naso-pharyngo-laryngoscopy and a chest x-ray. Other investigations such as a skin prick test or a RAST test may be tailored depending on clinical examination. Patients with suspected asthma are advised to keep a peak flow diary and record their findings twice daily for a fortnight.
The goal of treatment includes both symptomatic relief and treatment of underlying cause.
References
1. Millqvist E, Bende M. Role of the upper airways in patients with chronic cough. Curr Opin Allergy Clin Immunol 2006;6(1):7-11.
2. Chung KF, Pavord ID. Prevalence, pathogenesis, and causes of chronic cough. Lancet 2008;371(9621):1364-74.
3. Chung KF. Chronic cough: future directions in chronic cough: mechanisms and antitussives. Chron Respir Dis 2007;4(3):159-65.
4. Birring SS. New concepts in the management of chronic cough. Pulm Pharmacol Ther 2011;24(3):334-8.
5. Birring SS. Controversies in the evaluation and management of chronic cough. Am J Respir Crit Care Med 2011;183(6):708-15.
6. Chung KF. Chronic ‘cough hypersensitivity syndrome’: a more precise label for chronic cough. Pulm Pharmacol Ther 2011;24(3):267-71.
7. Morice AH. Chronic cough hypersensitivity syndrome. Cough 2013;9:14.
8. Pratter MR, Bartter T, Akers S, DuBois J. An algorithmic approach to chronic cough. Ann Intern Med 1993;119(10):977-83.
9. Fokkens WJ, Lund VJ, Mullol J, et al. European position paper on rhinosinusitis and nasal polyps 2012: Rhinology suppl 2012;23:1-298.
10. Shao-Cheang Liu, Hsing-Won Wang, Wan-Fu Su. Endoscopic vidian neurectomy: the value of preoperative computed romographic guidance. Arch Otolaryngol Head Neck Surg 2010;136(6):595-602.
11. Morice AH. Epidemiology of cough. Pulmonary Parmacology and Therapeutics 2002;15:253-9.
12. O’Hara J, Jones NS. The aetiology of chronic cough: a review of current theories for the otorhinolaryngologist. J Laryngol Otol 2005;119(7):507-14.
13. Irwin RS. Chronic cough due to gastroesophageal reflux disease: ACCP evidence-based clinical practice guidelines. Chest 2006;129(1 Suppl):80S-94S.
14. Pratter MR, Brightling CE, Boulet LP, Irwin RS. An empiric integrative approach to the management of cough: ACCP evidence-based clinical practice guidelines. Chest 2006;129(1 Suppl):222S-231S.
15. Raherison C, Montaudon M, Stoll D, et al. How should nasal symptoms be investigated in asthma? A comparison of radiologic and endoscopic findings. Allergy 2004;59(8):821-6.
16. Irwin RS, Curley FJ, French CL. Chronic cough. The spectrum and frequency of causes, key components of the diagnostic evaluation, and outcome of specific therapy. Am Rev Respir Dis 1990;141:640-7.
Acknowledgement
We would like to thank and acknowledge Professor AH Morice, Respiratory Physician, Hull University, UK for his guidance.
Declaration of competing interests: None declared.