Patrick Bradley ruminates on a celebrated career in ENT head and neck surgery and suggests that increasing the possibility of positive outcomes to contemporary patient safety initiatives by the NHS must involve efforts to develop an enthusiastic contented workforce willing to actively support these changes, which ultimately will also lead to reducing the medical negligence bill.
Introduction
The annual cost to the English National Health Service of settling clinical negligence claims is equivalent to training 6,500 doctors, (current cost £230,000 each) and is expected to double by 2023 according to the Medical Protection Society Study in 2017 [1]. A figure of £1.7bn was spent in 2017, reflecting a doubling of the annual cost since 20010/11. NHS Resolution has already estimated that £65 billion, up from £29 billion in 2014/15, will be needed for future clinical negligence costs – for claims arising from incidence that have already occurred [2].
Patient safety has been high on the national and international agenda in healthcare for almost a decade. In the UK reviews of case records have shown that over 10% of patients experience an adverse event while in hospital; similar figures have been reported from studies around the world. The question to be asked, after considerable investment time, effort and money is, “are patients any safer?” Some of the aspects of safety are difficult to measure for technical reasons (defining preventable), the main problem is that measurement and evaluation have not been high on the agenda. It is believed that the lack of reliable information on safety and quality of care is hindering improvement in safety across the world [3]. Measuring safety in healthcare is much more difficult than measuring safety in other domains, where mistakes and injuries are fewer, less varied, and can be more clearly defined [4].
“In the majority of medical negligence investigations, claimants and family members say that they are not interested in financial compensation for their injury, more that they wish the doctor in question to be held accountable.”
NHS healthcare safety
In the NHS the National Patient Safety Agency (NPSA) was established in 2001 to monitor patient safety incidences, including medications and prescribing error reporting. The NSPA developed a National Reporting and Learning System (NRLS) to collect and analyse information from staff and patients, as well as incorporating information from other sources. In 2012, the key functions of the NPSA were transferred to the NHS Commissioning Board Special Health Authority, later known as NHS England. In April 2016 the patient safety function was transferred from NHS England to the newly established body called NHS Improvement. In 2017 the Healthcare Safety Investigation Branch (HSIB) was set up, and while HSIB is funded by the Department of Health and hosted by NHS Improvement, this body operates independently of them and other organisations such as the Care Quality Commission (CQC). The current process is under review, yet again, with a call for responses closing in June 2018, with the aim of redrafting the Serious Incidence Framework 2015/16. With the launch of the HSIB it has been remarked that attempts to improve patients’ safety will result in creating a safety culture and make the NHS a learning culture.
What is required is a fundamental rethink on everything related to healthcare, from clinical training and pathway design, to culture and skills of trust boards, the inspection regime, and the regulation of organisations and professions. The best safety culture comes from the bottom-up, rather than the top-down [5].
Classification of patient adverse events
A working classification of ‘Patients’ Adverse Events’ is commented upon within the NHS ‘Serious Incidence Framework’ 2015/16 (Table 1). All such events should be identified, recorded and investigated. Almost annually the NHS ‘Never Events List’ has been added to and currently some 25 events are listed. The NHS has introduced financial measures to penalise service providers when these events occur – it is unsure how these events will be investigated and how others will learn when a never event occurs [6]. ‘Near misses’ are indistinguishable from ‘adverse events’ in all but the outcome, and are viewed as opportunities for quality improvement. It has been estimated that near misses occur 300 times more often than adverse events and typically precede a related adverse event (Figure 1), however most are not recorded or reported [7]. Safety in modern healthcare is a constantly moving target, as innovation and improving standards in healthcare alter our conceptions of both harm and preventability [8].
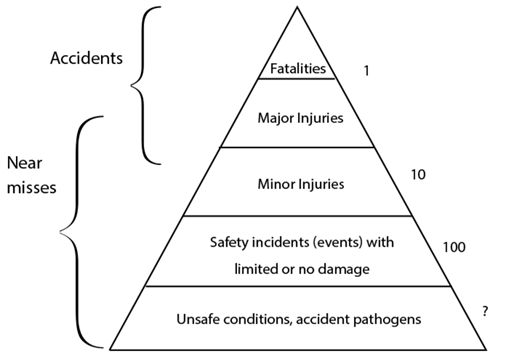
Figure 1: A safety pyramid.
Comments on current NHS practice
During my 27-year career as a consultant head and neck oncologic surgeon, I had only one attempt at a liability claim (claim collapsed on expert advice) and one letter from the General Medical Council regarding ‘fitness to practise’. I may have been lucky, or some would say unfortunate! Getting such mail can have a terrifying effect on one’s confidence, ability and possibly make one reflect on one’s future clinical practice. Complaints then (and probably now) were handled in the NHS by a manager, usually in great haste, frequently without gaining accurate facts, and offering patients an opportunity of meeting with the accused without their agreement! Such a process needs to be seriously changed and supervised by a senior clinician who is responsible for audit and clinical risk.
The British Medical Association (BMA) published a paper on the impact of complaints procedures on the welfare, health and clinical practice of 7926 doctors in the UK, and doctors’ perceptions of support and the process involved in complaints investigations [9]. One of the conclusions was that morbidity is greatest in cases involving the GMC [10]. The GMC’s guidance on ‘Good Medical Practice’ (March 2013, updated in 2014) gives basic information for all doctors, irrespective of their specialty and all seem obvious – sadly these have to be written and circulated, and are a source for the legal profession should patients consider that they have been mistreated or injured.
It has been recorded that in the majority of medical negligence investigations, claimants and family members say that they are not interested in financial compensation for their injury, more that they wish the doctor in question to be held accountable.
Misdiagnosis and delayed diagnosis
One area that always troubled me during my time as a clinician was the ‘two-week referral’ process for early diagnosis of cancer – more so with head and neck cancer. The process matured from a list of ‘red flag symptoms’ first devised by specialists (secondary care), followed by revisions by specialists (primary care) which reallocated some of the cardinal symptoms to neighbouring hospital specialists. According to the referral guidelines that less than 10% of referrals were diagnosed with cancer. It has been reported that >70% conformed to the recommended guidelines and had a higher positive predictability for cancer – 12.8%, whereas those patients not complying to the recommended referral guidelines had a lower positive predictability for cancer – 6.2% [11].
While this urgent referral of patients with symptoms continues, consultant clinicians specialising in the treatment of head and neck cancer have delegated such a screening clinic to non-consultant training staff. It would be interesting to know how many patients have been reassured and ultimately were subsequently diagnosed with a head and neck cancer.
This perception and aspiration is admirable but as a head and neck oncologic surgeon, the decision to treat and more so carry out surgery is made and supported by members of the multidisciplinary team (MDT), most of whom are not surgeons. Unfortunately when the surgery performed has not resulted in the expected outcome, it is the name over the bed that is remembered and blame is allocated to the named surgeon. Surgeons need to be more aware of this risk than the other non-surgical members of the MDT.
“Patient satisfaction and training must both be recognised and prioritised by the healthcare provider, which must over-ride cost savings and efficiency.”
Loss of team practice
The ‘team approach’ with leadership provided by the consultant has been eroded by the introduction of the European Working Time Directive (EWTD). To make meaningful gains in safety and continuity of patient care, a solution is to increase the numbers of doctors substantially, trained either at home or abroad [12]. Surgery skill and confidence are learned through experience – haltingly and humiliatingly. In surgery there remains a conflict between imperative to give patients the best possible care and the need for novices to gain experience. No matter how many safeguards are put in place, on average teaching cases go less well with trainee or even ‘new’ consultants than with someone experienced. A report by the Royal College of Surgeons of Edinburgh highlights the inefficiencies in front line surgical services that make it harder to deliver a safer service and that lead to poor communication, high stress levels and limited training opportunities [13]. The summary states that: “the lack of team structures, insufficient handovers, inadequate time for training, and reduced support from senior colleagues individually are significant concerns, but the combined effect of these factors could compromise surgical safety”. There is a price to pay, by patients and service provision, to educate future surgeons for the NHS – patient satisfaction and training must both be recognised and prioritised by the healthcare provider, which must over-ride cost savings and efficiency.
“Surgeons should rise to the challenge of leading checklists in the operating room and be the champions of safety within their hospitals.”
WHO Surgical Safety Checklist
In 2007 the World Health Organization (WHO) drafted a ‘Surgical Safety Checklist’ for briefings in the operating room. This involves interrupting the anaesthetist and the surgeon at three specific times during surgery so that information can be checked and communicated to all team members. These three times are immediately before commencing anaesthesia (sign-in) immediately before skin-incision (or endoscopy) (time-out) and immediately on completion of surgery skin-closure (sign-out) [14]. In the UK, universal implementation of the WHO checklist is officially required, but a study in England reported wide variation of usage with incomplete data and without the presence or focus of the entire surgical team. It was shown that senior surgical leadership of the checklist was associated with better checklist usage; surgeons should rise to the challenge of leading checklists in the operating room and be the champions of safety within their hospitals [15]. Furthermore, while operative diagnostic and therapeutic surgery is highlighted as requiring a ‘pre-flight checklist’ there are many similar clinical practices that need special attention to reduce adverse events.
Learning from lawsuits
Data from the NHS Litigation Authority (NHSLA) from 2004 to 2014 for 11 surgical specialties were reviewed [16]. Orthopaedic, obstetric and general surgery received the largest number of claims per year, and paediatric surgery the least. Otorhinolaryngology (ORL), oral and maxillofacial surgery (OMFS) and plastic surgery (PS) had less than 160 claims per year, which reflects the volume of finished consultant episodes per specialty over a year period. Neurosurgery was responsible for the highest average amount paid per claim, and OMFS the lowest.
The three leading types of claims were failure/delay in treatment and/or diagnosis and failure to warn/adequately consent. OMFS and PS both received a high volume of successful claims relating to failure to warn/inform consent. ‘Never events’ or unexpected deaths as primary claims included wrong side surgery, retained foreign body/instrument, or wrong implant/prosthesis (zero across all three specialties) over a 10 year period. The numbers for these are very small compared with the three major surgical disciplines.
Currently litigation associated with surgery accounts for 40% of all clinical negligence claims in England. Understanding where patient management has fallen short enables litigation and its significant burden to be minimised. The NHS has responded by supporting a national programme called ‘Getting it right first time’ (GIRFT), which received additional government funding in November 2016 of £60 million to expand and accelerate delivery [17]. The programme is composed of and lead by frontline clinicians, covering more than 32 specialties, and was created to help improve the quality of medical and clinical care within the NHS by identifying and reducing unwarranted variations in service and practice. Early reports have naturally concentrated on the major surgical disciplines, but with time and as data is accumulated, then reports on ORL, OMF and PS will follow. However an area in need of urgent attention is preoperative consent for commonly performed ORL operations such as grommet insertion, tonsillectomy, septoplasty and hemithyroidectomy which findings indicate is variable and does not reflect the operative risk. There is a need for a change and uniformity of consent documentation and information discussion, across all likely grades of clinicians if patient autonomy is to be protected, and the requirements of local laws and regulatory bodies met [18].
While ORL does not currently attract much litigation in the NHS, there is no reason to be complacent! During my time there have been many changes, but one particular procedure that was useful for teaching and cross clinical contacts was the grand rounds once a month, which included morbidity and mortality data supported by evidence, (in the case of mortality with autopsy findings). Should we reinstate these clinical gatherings?
Summary
The NHS has produced several initiatives over the past decade aimed at reducing the annual cost of patient litigation by involving staff, data collection, publication etc. Indeed it seems that newer initiatives are being developed or revised before the previous strategy has been bedded into clinical practice! There is a need for investment in improving the morale and well being of the workforce. This area has been sorely neglected by the current government and NHS with the constant target setting, while at the same time reducing funding, which has resulted in closure of facilities, non-replacement of equipment, and a “conscious reduction of staff levels”. Current NHS staffing is weary, angry and “squeezed dry of empathy”, which has resulted in a resistance to further or continued change. The key to change in the NHS as in other large organisations is a happy and contented workforce who will embrace change.
FURTHER READING
-
NHS Improvement. Patient Safety,
https://improvement.nhs.uk/
improvement-hub/patient-safety/ -
NHS Improvement. The future of NHS patient safety investigation, March 2018,
https://improvement.nhs.uk/documents/2525
The_future_of_NHS_patient_safety_investigations_
for_publication_proofed_5.pdf -
General Medical Council. Good Medical Practice, 2013,
www.gmc-uk.org/guidance -
Serious Incidence Framework 2015/16,
https://improvement.nhs.uk/
documents/920/serious-incidence framework.pdf -
NHS Improvement. Never Events, 2018,
https://improvement.nhs.uk/
documents/2266/Never_Events_list_2018_FINAL_v5.pdf -
Gawande A. Complications: A surgeon’s notes on an imperfect science. New York, USA; Henry Holt and Co; 2002.
-
Syed M. Black Box Thinking. Part 1: The Logic of Failure. London, UK; John Murray; 2016.
References
1. Medical Protection Society: Who pays the Price?
www.medicalprotection.org/balance
Last accessed August 2018.
2. NHS compensation payouts ‘unsustainable’, say health leaders. The Guardian.
www.theguardian.com/society/
2018/feb/02/nhs-compensation-payouts-
unsustainable-say-health-leaders
Last accessed August 2018.
3. Vincent C, Aylin P, Dean B, et al. Is health care getting safer? BMJ 2008;337.
4. Vincent C, Burnett S, Carthey J. Safety measures and monitoring in healthcare: a framework to guide clinical teams and healthcare organisations in maintaining safety. BMJ Qual Saf 2014;23(8):670-7.
5. The NHS needs a culture shift if it truly wants to put patient safety first. The Guardian.
www.theguardian.com/healthcare-network/
2017/jun/02/nhs-culture-shift-patient-safety-first
Last accessed August 2018.
6. Van Spall H, Kassam A, Tollerfson T T. Near-misses are an opportunity to improve patient safety: adapting strategies of high reliability organisations to healthcare. Curr Opin Otolaryngol Head Neck Surg 2015;23(4):292-7.
7. Gnoni M G, Saleh J H. Near-misses management systems and observational-in-depth: Handling safety incidences and accident precursors in light of safety principles. Safety Science 2017;91:154-67.
8. Vincent C, Amalberti R. Safety in healthcare is a moving target. BMJ Qual Scf 2015;24:539-40.
9. Bourne T, Wynants L, Peters M, et al. The impact of complaints procedures on the welfare, health and clinical practice of 7926 doctors in the UK: a cross-sectional survey. BMJ Open 2015;4:e006687.
10. Bourne T, De Cock B, Wynants L, et al. Doctors’ perception of support and the process in complaints investigations and how these relate to welfare and defensive practice: a cross-sectional survey of the UK physicians. BMJ Open 2017;7:e017856.
11. McKie C, Ahmed U A, Fellows S, et al. The 2-week rule for suspected head and neck cancer in the United Kingdom: referral patterns, diagnostic efficacy of the guidelines and compliance. Oral Oncol 2008;44(9):851-6.
12. Goddard A F. Goodbye to the European Working Time Directive? Editorial BMJ 2016;354:13702.
13. Improving the working environment for safe surgical care. Royal College of Surgeons of Edinburgh.
www.rcsed.ac.uk/media/
415574/rcsed-working-
environment-web3.pdf
Last accessed August 2018.
14. Fudickar A, Horle K, Wiltfang J, Bein B. The effect of the WHO Surgical Safety Checklist on Complication Rate and Communication. Dtsch Arztebl Int 29912;109(42):695-701.
15. Russ S, Roout S, Caris J, et al. Measuring variation in use of the WHO Surgical Safety Checklist in the operating room: a multicenter prospective cross-sectional study. J Am Coll Surg 2015;220:1-11.
16. Ford K E, Cooper L R L. Learning from lawsuits: Ten-years of NHS litigation authority claims against 11 surgical specialties in England. The Surgeon 2018;16:27-35.
17. The Getting it Right First Time Programme: Early views from the provider sector. NHS Providers.
http://nhsproviders.org/media/
4365/girft-final-pdf.pdf
Last accessed August 2018.
18. Smith M E, Lakhani R, Bhat N. Consenting for risk in common ENT operations: an evidence-based approach. Eur Arch Otorhinolaryngol 2013;270:2551-7.





