Why are we developing this technology?
A key problem in drug delivery is getting the therapy to the right place in the body, which is especially challenging for targets that are small, deep and are protected or surrounded by anatomical barriers. We have shown that magnetic forces can be used to minimally-invasively direct therapy to middle and inner ears [1-5]. Using iron-core particles and a ‘magnetic injector’, drug-coated nano-particles have been transported from the outer to the middle ear (through the tympanic membrane), or from the middle to the inner ear (through the round and oval window membranes).
After magnetic delivery, the particles elute drugs at a controlled rate to provide a therapeutic effect. For middle ear conditions, such as middle ear infections, this could allow delivery of therapy into middle ears without ear drum puncture or general anaesthesia in children. For conditions that originate in the inner ear, such as sudden sensorineural hearing loss (SSNHL), Ménière’s, and tinnitus, pre-clinical experiments have shown that drug delivery to the cochlea can be increased substantially over passive diffusion mechanisms.
What treatments are in use at present? Which patients could benefit from this technology?
For middle ear infections such as acute otitis media (AOM), the standard-of-care is usually systemic antibiotic administration [6,7]. Only a small fraction of the administered drug reaches the middle ear. There are no effective non-invasive treatments for chronic otitis media with effusion (COME), nor are there medical treatments that reliably block the progression of AOM to COME. For this reason, tympanostomy tube placement under general anaesthesia for treatment of recurrent AOM or COME is the most common paediatric surgical procedure requiring general anaesthesia in the United States [8]. Magnetic trans-tympanic drug delivery in children, without pain or general anaesthesia and its attendant risks, could go a long way towards improving the treatment of children with AOM or COME.
The current standard-of-care for SSNHL, an inner ear indication, often involves injection of a steroid solution through the tympanic membrane, partially filling the patient’s middle ear. The patient then remains in a supine position for up to an hour to allow the drug to permeate into the inner ear. This method relies on passive diffusion of the drug from the middle ear into the cochlea, but only a limited amount of the drug reaches even the basal turn of the cochlea, and it is likely that virtually no drug reaches the apex [9-11]. The resulting efficacy of this approach is similar to orally administered steroids [10]. Overall, treatment efficacy for SSNHL under the current standard-of-care is uncertain [12, 13]. In contrast to passive diffusion of drug from the middle into the inner ear, magnetic forces actively transport therapy into the cochlea.
At what stage of development are the technology and the commercialisation effort?
We are at an early stage. We have an invention, key patents granted to protect it, an optimised magnetic injection system, pre-clinical animal data, and a team that combines engineering, medical and business expertise. Otomagnetics, our start-up company, was formed in March 2012. Over the last year and a half, we have secured non-dilutive funding from the Maryland Industrial Partnerships (MIPS) programme, from the Maryland Innovation Initiative at the Maryland Technology Development Corporation (TEDCO), from Action on Hearing Loss in the UK, as well as from the Sheikh Zayed Institute for Pediatric Surgical Innovation, Children’s National Medical Center, in Washington, DC. This support is enabling us to conduct needed animal safety and efficacy studies, as well as to expand our patent portfolio and develop our business approach. The company’s magnetic delivery technology will require regulatory approval before it can reach patients, thus we are currently pursuing investor funding and are applying for further research and business development grants to fund the needed product development and testing.
What kind of data do we have so far?
In our pre-clinical rat middle ear studies, we showed that 100 nm diameter particles were able to traverse the ear drum. We placed the particles into the ear canal and applied the magnet at a 3 cm distance from the ear drum (to match the maximum distance anticipated in human children). Magnetic forces were able to deliver therapeutic amounts of drug into the middle ear, without a need for ear drum puncture.
For inner-ear experiments we deposited a solution of 300 nm diameter prednisolone-coated chitosan-matrix iron-core particles into rat middle ears via intra-tympanic injection, and then held our magnet device at a 4 cm distance from the rat’s cochlea (to match the anticipated distance from magnet to cochlea in adult humans). We observed that magnetic delivery improved both the amount (by a factor of ten) and the uniformity of drug (a factor of three increase in apical-to-basal concentration) delivered to the cochlea. Drug uniformity was improved because magnetic forces direct drug-coated particles to a substantial portion of the cochlea. In behavioural studies, we showed that magnetic injection could prevent tinnitus following noise trauma, and reduce noise-induced hearing loss compared to trans-tympanic injection of free drug into the middle ear [2, 14]. We also validated, by auditory brainstem response (ABR) and by behavioural audiogram studies, that magnetic delivery of drug-coated nano-particles into the cochlea did not harm hearing in healthy animals [4, 15].
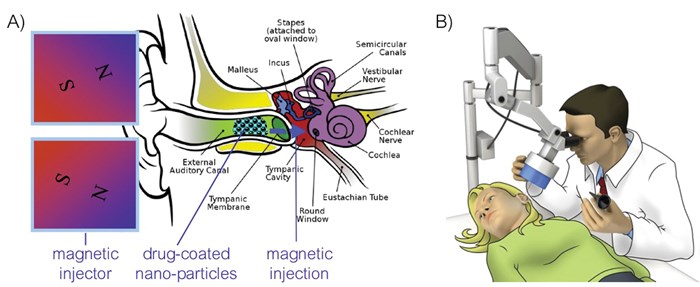
Figure 1: A) Illustration for minimally-invasive drug delivery into the middle ear, without a need to puncture the ear drum. B) Intended clinical use, the magnetic injector is shown in light blue.
What is the product we hope to bring to market?
To better treat patients, we envision a product that is an easy-to-use magnetic injector and a therapeutic drug / particle formulation, to be used as shown schematically in Figure 1. For middle ear infections, we seek to reduce the reliance on tympanostomy tubes which require surgery and general anaesthesia with its attendant costs and risks in children. Our goal is to improve outcomes and reduce overall costs with a simple, in-office, non-invasive procedure. A clinician would place a solution or gel containing drug-coated particles into the ear canal, and then would use our magnetic injector to direct the therapy into the middle ear, without surgery, anaesthesia, or the usual post operative potential complications. We believe such a minimally-invasive treatment for AOM or COME would be welcomed by doctors, paediatric patients and their parents.
For conditions that originate in the inner ear, such as SSNHL, Ménière’s and tinnitus, the clinician would trans-tympanically inject drug-eluting magnetic particles and would then use the magnet device to effectively direct them into the cochlea, improving drug dose and uniformity. This could address the problem encountered by clinicians of insufficient and non-uniform drug delivery to the cochlea, which has been implicated in patient-to-patient variability and lack of efficacy [12, 13].
How did we determine that this is a commercially viable product?
The middle ear market is substantial. In the United States, there are over 15 million cases per year of acute (early-stage) and chronic (persistent, with mucous build up) middle ear infections in children under five years of age [16]. If 10% of those cases were treated with magnetic drug delivery, at a fraction of the cost of a tympanostomy tube procedure (>$1500 after facility, anaesthesia and surgery costs), that alone would be a half billion dollar US market. A similar large opportunity exists in Europe. Minimally-invasive magnetic injection into the inner ear, as well as to other targets (eyes, skin), presents additional market opportunities, ranging from small markets for orphan indications (e.g. for SSNHL) to substantial markets for magnetic drug delivery for more common diseases (tinnitus, presbycusis, macular degeneration, and topical drug delivery to non-healing wounds).
What are the next challenges? And how do we plan to overcome them?
Bringing the technology to market will require extensive animal and clinical testing, as necessary for regulatory approval, and will likely take 5-10 years and tens of millions of dollars. To reduce risk and time-to-market, our goal is to first pursue an adult indication that enables fast patient recruitment (such as SSNHL, which is considered an otologic emergency), while in parallel collecting safety and efficacy data to approach the larger otitis media paediatric market. We have been successful at securing substantial non-dilutive funding, and are partnering with Children’s National Medical Center in the US and Nottingham University Hospitals in the UK to develop and test the technology, and in the future to recruit patients for clinical trials. We are also pursuing partnerships with pharmaceutical companies (one pilot project is already in place) to deliver their therapies to middle and inner ear targets. We believe this technology will eventually be used widely by otolaryngologists, but as with any disruptive innovation, adoption will require time and money to achieve proof of efficacy, safety and economic benefits to patients and doctors.
Acknowledgements
Mika Shimoji, David Beylin, Irving Weinberg and Ting Pau Oei, at the University of Maryland, Weinberg Medical Physics, and Fox Feather Ventures respectively, contributed to the research and to business development for this technology. Funding support is gratefully acknowledged from Action on Hearing Loss in the UK, from the National Institutes of Health (NIH) in the USA, and from the Maryland Innovation Initiative at the Maryland Technology Development Corporation (TEDCO) and the Maryland Industrial Partnerships (MIPS) programme in the State of Maryland, as well as from the Sheikh Zayed Institute for Pediatric Surgical Innovation, Children’s National Medical Center, in Washington DC.
References
1. Sarwar A, Lee R, Depireux DA, Shapiro B. Magnetic Injection of Nanoparticles into Rat Inner Ears at a Human Head Working Distance. IEEE T Magn 2013;49:440-52.
2. Sarwar A, Depireux D, Basiri R, Lee R, Shapiro B, Weinberg I. Magnetic Nano-Particles to Deliver Drugs to Ears. Mid-winter meeting of the Associate for Research in Otolaryngology, Baltimore, MD; 2013.
3. Sarwar A, Lee R, Depireux D, Shapiro B. Treating Tinnitus by Magnetic Pushing of Therapy: Rat Experiments. 9th International Conference on the Scientific and Clinical Applications of Magnetic Carriers, Minneapolis, MN; 2012.
4. Depireux DA, Lee R, Sarwar A, Shapiro B. Drug delivery to the inner ear of rats using magnetically steered nanoparticles. Society for Neuroscience Annual Meeting, Washington, DC; 2011.
5. Shapiro B, Kulkarni S, Nacev A, Sarwar A, Depireux D. Shaping Magnetic Fields to Direct Therapy to Ears and Eyes. Annu Rev Biomed Eng 2014; accepted.
6. American Academy of Pediatrics (AAoP). American Academy of Pediatrics Subcommittee, diagnosis and management of acute otitis media. Pediatrics 2004;113:1451-65.
7. Hoberman A, Paradise JL, Rockette HE, et al. Treatment of acute otitis media in children under 2 years of age. N Engl J Med 2011;364:105-15.
8. Kogan MD, Overpeck MD, Hoffman HJ, Casselbrant ML. Factors associated with tympanostomy tube insertion among preschool-aged children in the United States. Am J Public Health 2000;90:245-50.
9. Haynes DS, O’Malley M, Cohen S, Watford K, Labadie RF. Intratympanic dexamethasone for sudden sensorineural hearing loss after failure of systemic therapy. Laryngoscope 2007;117:3-15.
10. Bird PA, Murray DP, Zhang M, Begg EJ. Intratympanic versus intravenous delivery of dexamethasone and dexamethasone sodium phosphate to cochlear perilymph. Otology & Neurotology 2011;32:933-6.
11. Oertel R, Kirch W, Klemm E. Prednisolone concentration in the cochlea of patients with perilymph fistula. Pharmazie 2007;62:239-40.
12. Stachler RJ, Chandrasekhar SS, Archer SM, Rosenfeld RM, Schwartz SR, Barrs DM. Clinical Practice Guide: Sudden Hearing Loss. Otolaryngol Head Neck Surg 2012;146(S3):S1-35.
13. Conlin AE, Parnes LS. Treatment of Sudden Sensorineural Hearing Loss: II. A Meta-Analysis. Arch Otalayrngol Head Neck Surg 2007;133:582-6.
14. Depireux DA, Shapiro B. Preventing the emergence of tinnitus post-trauma with drug-loaded nanoparticles. 6th International Conference on Tinnitus, Bruges; 2012.
15. Sarwar A, Basiri R, Ahmed M, Lee R, Weinberg IN, Shapiro B, Depireux D. Magnetic Injection of Steroids into the Inner Ear Mitigates Acute Hearing Loss in Rats. Hearing Res 2014; in prep.
16. Grubb MS, Spaugh DC. Treatment failure, recurrence, and antibiotic prescription rates for different acute otitis media treatment methods. Clin Pediatr (Phila) 2010;49:970-5.
Declaration of Competing Interests
BS,DD, AN and JH are involved with the start-up company Otomagnetics. At present, BS, DD and AN have a financial stake in the company.