Tinnitus is a condition that affects over 300 million people worldwide. Typically it manifests as a ringing or buzzing in the ears and while there is not yet a cure there are many ways for patients to manage their tinnitus. Sound Relief sound therapy, by Restored Hearing, is one such way of managing tinnitus. Company founder Eimear O’ Carroll spoke to us about the new app and the benefit it brings for patients and clinicians alike.
What is Sound Relief?
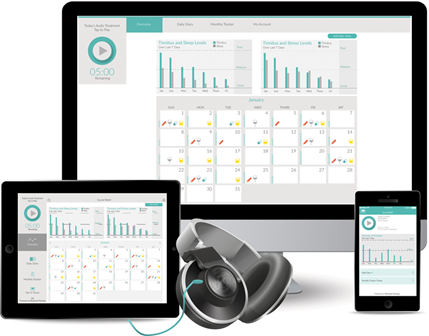
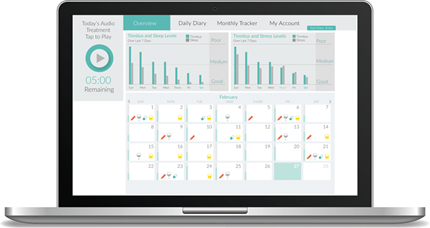
Sound Relief is an online sound therapy for tinnitus. It uses low frequency sound to alleviate tinnitus when used on a daily basis for 5-30 minutes. Sound Relief can be downloaded from the Apple App Store or accessed at RestoredHearing.com through any internet enabled device, where the sound therapy is streamed using any normal pair of headphones. In addition to the therapy, there are tinnitus tracking functions to enable the patient to monitor the factors that may influence their tinnitus and their Tinnitus Handicap Inventory (THI) score over the course of treatment. These results can then be shared and discussed with their clinician for further analysis.
What’s the evidence for using Sound Relief for tinnitus?
Sound Relief was tested in a randomised controlled trial of 300 people at the University of Edinburgh. Around two thirds of tinnitus patients received a benefit from using the therapy for five minutes per day over a 30 day period, where the average benefit was a 25% reduction in tinnitus severity as measured by the THI and Tinnitus Functional Index (TFI). So far the app has been a very successful tool for patients in managing their tinnitus, with many subscribing for months on end. Further studies are planned for later in 2016 to examine the longer term effects.
As a clinician, which of my patients would be suited to Sound Relief?
It works for subjective tinnitus patients. They should have already had their tinnitus assessed by a clinician and be considering non-invasive treatment options. Sound Relief is recommended for adults only and the trials took place on those experiencing tinnitus for between two and 15 years.
What are the benefits of Sound Relief for the patient?
The 30 day free trial allows tinnitus patients to try Sound Relief at no risk before subscribing. Sound Relief is non-invasive, easy to access, and an affordable option for patients in the management of tinnitus. As mentioned before, there’s an average 25% reduction in the first month of use for the two thirds of patients who see a benefit. Additionally, the tracking functionality provides a simple interface for gaining a greater understanding of one’s tinnitus, which can be of immense comfort in its own right.
What do your current patients say about Sound Relief?
As with any tinnitus therapy, there are patients for whom the treatment won’t work. The feedback, however, has been positive on the whole. For example, Mark from the Netherlands said, “The free trial was a great way to be introduced to your sound therapy. It has been a very positive experience for me which means I’ll definitely continue with Sound Relief in the hopes of further reducing my tinnitus.”
How does the patient access the treatment?
It’s all online so any device that gets a patient online will allow them access to Sound Relief. Patients can either go to RestoredHearing.com or download Sound Relief from the Apple App Store. Once they’ve signed up using an email address – no credit card details required – they plug in their own headphones and start their 30 day free trial of the sound therapy. The tracking functionality of Sound Relief is free forever.
“So far the app has been a very successful tool for patients in managing their tinnitus.”
As a clinician, why should I recommend Sound Relief to my patients?
Sound Relief sound therapy is a low risk and easy to use option that clinicians can offer their tinnitus patients. No credit card details are required for the 30 day free trial so patients can be quickly signed up in clinic or at home. Monthly THI and daily diary tracking provide more detailed information for clinicians about their patients’ tinnitus and factors that may be influencing it. This allows for more constructive discussions during patient appointments.
You’ve mentioned new tracking functionality, how does that work?
There are two types of tracker available – a daily diary and a monthly THI. The daily diary provides a short form to record daily consumption of alcohol, medication, and caffeine. There are also sliding scale measures of tinnitus levels which are then compared with sleep quality and stress levels. Charts present the entered data in an easy to understand graphic format. Once a month patients can complete the THI, again using sliding scales, and results are displayed in a 12 month graph. This is particularly useful for clinicians in monitoring their patient’s tinnitus over time.
Can my patients share their results with me?
Yes, THI results can be emailed by the patient to their clinician, friends, or family. It’s hoped that this information will promote a more informed discussion when a patient comes in for their appointments. Sharing functionality for the daily diary will come later in 2016.
How much does Sound Relief cost and who pays?
Patients are in control of their accounts, payment details, and data. Tracking functions on the app are always free, only an account is required. After the 30 day free trial is completed subscription plans start at $29.95 per month. Patients can pause their subscriptions easily with no long-term commitments required.
Are there any benefits to clinicians for referring Sound Relief?
Sound Relief offers a straightforward therapy option that can be offered to tinnitus patients where it is deemed suitable by their clinician. We do provide extra benefits for trusted partner clinics and audiologists who refer Sound Relief to their patients. If you deal with tinnitus patients and are interested in signing up to our referral programme, please contact us at https://restoredhearing.com.