Berthold Langguth describes an innovative project that is creating the world’s largest multi-national patient database containing standardised information on tinnitus and medical history, otological examination and psycho-acoustic measures of tinnitus.
A challenge for tinnitus treatment and tinnitus research is the identification of the most promising therapy for a specific patient. One approach to address this challenge is the systematic assessment of tinnitus patients in many clinical centres with standardised instruments (e.g. psychoacoustic measures, questionnaires). For this purpose the Tinnitus Research Initiative (TRI) database has been created, which is further developed and enlarged in the current COST Action TINNET.
The Tinnitus Patient Database was started in 2008 as a project of the Tinnitus Research Initiative (TRI). Standardised methods for tinnitus patient assessment and treatment outcome measurement were chosen according to a consensus reached by tinnitus experts from many countries during an international tinnitus conference in Regensburg in 2006. These core assessments consist of a standardised tinnitus and medical history, otological examination, psycho-acoustic measures of tinnitus and a variety of validated questionnaires assessing tinnitus severity and quality of life.
“Clinical and research centres from all over the world can contribute to the database.”
All participating centres are supplied with standardised case report forms (CRF) containing these items. The design of the CRF allows the documentation of both cross-sectional and longitudinal data. CRFs are currently available in English, French, German, Spanish, Italian, Czech, Flemish, Persian, Polish, Portuguese and Swedish. Translations in further languages are in preparation. Twenty-eight centres from 16 different countries are currently contributing to the database and the number of contributing centres is continuously growing.
The Database Working Group represents the backbone of the TINNET project as it connects the activities of the other four workgroups. The multidisciplinary working group consists of clinicians, clinical trial specialists, data documentation specialists, software engineers, biostatisticians and mathematicians.
“The database is a resource for all clinicians and researchers in the tinnitus field.”
The goal of the Database Working Group is to provide the technical, legal and administrative framework that enables the integration of data from clinical patient assessment (WG1), neuroimaging (WG3), genetic analysis (WG4) and outcome measurement (WG5). Current standards for data collection are updated and methodologically improved in the TINNET COST Action by WG1 (clinical) and WG5 (outcome measurements). Moreover the database structure will be enlarged to enable interaction with information from neuroimaging and genetic studies. The database structure will allow updates and upgrades of data standards, which ensures that the database can be further developed according to newest scientific standards. With these activities the database is expected to further grow (Figure 1).
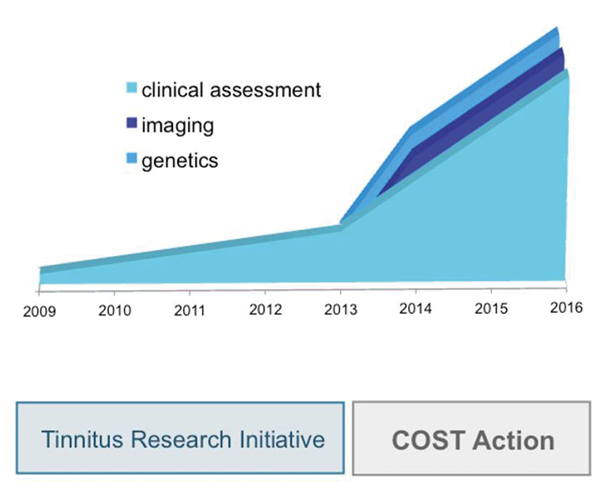
Figure 1: Development of the TRI Database within the TINNET COST Action.
Major topics within the WG3’s activities are the establishment of standards for collaboration within the database network, standards for data handling and data management and standards and strategies for data analyses. Further important issues which are addressed include legal and regulatory aspects, securing of data safety and privacy, scientific and administrative agreements and publication policies. A particular focus is on a user-friendly approach, to enable data collection in clinical routine treatment.
The goal is to provide technical support which both facilitates the clinical management of tinnitus patients and enables systematic data collection. As an example, patients should be able to complete standard questionnaires on a tablet computer while they sit in the waiting room of the doctor’s office. When they enter the doctor’s office, the entered data (scores of the various questionnaires) and their changes over time are displayed on a user-friendly interface. This information can be directly used in the communication with the patient (e.g. as basis for counselling) and for rational therapeutic decisions. Finally there will be standardised statistical analysis tools, which enable the clinician to evaluate therapeutic effects of larger patient cohorts (Figure 2).
Figure 2: Screenshot of the patient’s menu for entering the Tinnitus Handicap Inventory.
The TRI database is unique as it is the first database which collects prospective, standardised data from patients undergoing different types of treatment interventions. Clinical and research centres from all over the world can contribute to the database, if they are willing to adhere to the standards for patient assessment and outcome measurement. These standards are based on a consensus conference of tinnitus researchers and are currently being updated in the TINNET project. Data entered in the database range from highly selected patient populations participating in controlled trials to unselected patient populations receiving standard treatments under real world conditions. Thus, this database enables comparability and meta-analysis of study data from different clinical trials, different centres and from patient populations with differing ethnic backgrounds. The database has been constructed in a flexible modular structure, so that additional instruments can be included at any time in the future.
References
1. Landgrebe M, Zeman F, Koller M, et al. The Tinnitus Research Initiative (TRI) database: a new approach for delineation of tinnitus subtypes and generation of predictors for treatment outcome. BMC Medical Informatics and Decision Making 2010;10:42.
2. Meikle MB. Electronic access to tinnitus data: the Oregon Tinnitus Data Archive. Otolaryngology Head and Neck Surgery 1997;117(6):698-700.
3. Zeman F, Koller M, Schecklmann M, et al. TRI database study group. Tinnitus assessment by means of standardized self-report questionnaires: psychometric properties of the Tinnitus Questionnaire (TQ), the Tinnitus Handicap Inventory (THI), and their short versions in an international and multi-lingual sample. Health and Quality of Life Outcomes 2012;10:128.
4. http://www.tinnitusresearch.org
5. http://tinnet.tinnitusresearch.net/
images/pdf/WG2/database_report_11-02-15.pdf
Declaration of competing interests: None declared.
ABOUT THE AUTHOR
After studying medicine in Munich, Berthold completed residencies in neurology, psychiatry and psychotherapy. His tinnitus research interests span neuroimaging brain stimulation, pharmacological treatment and clinical trial methodology. Berthold proposed the COST Action initiative, represents Germany on the TINNET Management Committee and is chair of WG2.
Value of the database for tinnitus research
-
Collection of epidemiological data
-
Identification of candidate clinical characteristics for delineating neurobiologically distinct forms of tinnitus
-
Clinical phenotyping of tinnitus patients for neuroimaging and genetic research
-
Profiling of tinnitus patients based on their specific symptoms and / or their response to treatment modalities
-
Delineation of subgroups with similar characteristics and generating data about discriminative power of diagnostic procedures
-
Assessment of treatment outcome for specific treatments
-
Comparability of study results from different clinical trials, from different centres as well as from patient populations with differing ethnic backgrounds
-
Explanation of discrepant results from different studies (e.g. caused by the possibility of identifying differences between the study populations)
-
Identifying predictors for treatment response to specific treatments
-
Cross-validation of different assessment instruments in different languages
-
Development of an individualised treatment algorithm for every single patient based on the individual diagnostic profile.
Value for participating clinics
-
Visualisation of patient data at clinic visits
-
Visualisation of change over time e.g. under treatment
-
Possibility to compare own patients with the whole collective of tinnitus patients
-
Standard statistical analysis and visualisation of own patient data
-
Easy creation of an individualised case report form (CRF)
-
Individualised treatment recommendations based on the database.
How to participate in the database project
Every clinical and research centre that adheres to the standards for assessment and outcome measurement can participate in the database project.
Participation in the database project involves the following steps:
-
Contact with database office
-
Signature of the scientific agreement
-
Allocation of a centre ID
-
Data contribution via Internet
-
Continuous access to own data
-
Access to all data according to agreed database regulations
SUMMARY
-
The Tinnitus Patient Database is a large multi-national and rapidly growing patient database project, in which every interested clinician and researcher can participate.
-
Participation in the database project gives the clinician a fast and comprehensive overview of patients’ clinical characteristics and changes under treatment and thus improves the quality of patient management.
-
The large multi-national cross-sectional and longitudinal database is a unique resource for profiling of tinnitus patients and identifying outcome predictors for various therapeutic approaches.
-
By integrating data from clinical WG1, neuroimaging WG3, genetics WG4, and outcome measurements WG5, the Tinnitus Patient Database represents the backbone of the TINNET COST Action BM1306.




