So what has changed in four decades of OAEs? Do we now have all the answers? Have we reached our optimum recording ability? Professor Kemp explains what we know, what we don’t know and what’s to come.
In the 40 years since the surprizing discovery that ears make sounds, otoacoustic emissions (OAEs) have become commonplace in newborn hearing screening programmes. They have a role in the clinic as a supplementary test to clarify diagnoses and to record cochlear changes. (Figure 1). They continue to be a productive auditory research tool. More applications are to come.
Figure 1. Otoacoustic emissions are used every day for well-baby hearing screening,
and for clinical investigations, but they can do much more.
During those forty years we’ve learnt so much more about the cochlea and hearing loss that it’s appropriate to ask the questions - do we now completely understand OAEs and are we using them to best effect?
Gaining a thorough understanding of how the cochlea works is taking much longer than understanding the eye. Why? The cochlea is not just inaccessible it is also not just a sense organ – it’s a complex micro-machine that manipulates sound vibrations to prepare them for transfer to the auditory nerve. Of course the eye manipulates light for the same reason, but it focuses light with a single lens that we can see, examine, emulate and easily understand. Not so the cochlea!
“Gaining an understanding of the complex chain of events between middle ear and auditory nerve has taken decades, and research continues.”
The excitement caused by the discovery of otoacoustic emissions was not just because of the novelty of recording sound coming from an ear. It was because OAEs allowed measurements previously only possible invasively in the laboratory, to be made in the clinic. OAEs promised direct communication with the otherwise inaccessible ‘engine’ of cochlea (Figure 2). That ‘engine’ was a mystery in 1978 but only five years later another discovery began to resolve the mystery. Outer hair cells (OHCs) possessed ‘electro-motility’. They ‘twitched’ when they received sound vibration, an action capable of changing the way the whole cochlea responded to sound. Electromotility determines what vibrations reach the sensory inner hair cells (IHCs) and what information the IHCs can pass to auditory nerve fibres. It also is responsible for OAEs.
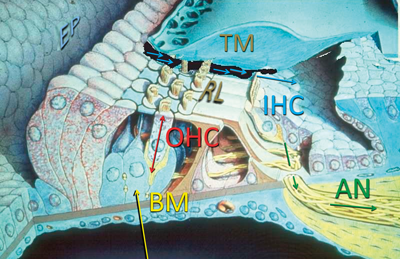
Figure 2. The ‘engine of the cochlea’ – which determines hearing quality and sensitivity is also responsible for OAEs. The organ of Corti is a micro-machine that converts up/down basilar membrane (BM) motion into horizontal fluid motion between reticular lamina (RL) and tectorial membrane (TM) deflecting the stereocilia of the inner hair cells (IHC) which then triggers auditory nerve (AN) spikes. The mechanism is power-assisted by outer hair cell (OHC) electro-motility, drawing energy from the endocochlear potential (EP). OAEs arise when this energy escapes. (Artwork courtesy of Martin Robinette, Mayo Clinic.)
Gaining an understanding of the complex chain of events between middle ear and auditory nerve has taken decades, and research continues. But it has been clear since the 1980s that OHCs’ transformation of the cochlea’s physical response to sound vibration is crucial for normal hearing. OHCs are also the most vulnerable mechanism in the auditory system and the vast majority of sensory deafness cases involve OHC dysfunction and lowered OAE production. This is the core reason why we need OAEs in the clinic and why OAEs are so effective in well-baby hearing screening.
What are OAEs? OAEs are a functionless leakage of OHC ‘twitch energy’, back to the ear canal. Their strong presence is a good sign indicating that OHCs are working and in a normal environment. That in turn means IHCs will receive the correct pattern of vibration for interpretation by the brain, at least at the frequency being tested. It doesn’t prove IHCs or their afferent nerves are functioning so it’s not a hearing test.
So what exactly do OHCs do to sound vibrations in the cochlea to prepare them for neural processing – and how do OAEs reflect that process? OHCs add energy to the vibration initiated by sound. This compensates for energy that is naturally lost in tissue and fluid movement enabling the basilar membrane to do its job much better. Its role is to carry sound vibration along the cochlea while separating its component frequencies. It sharply ‘focuses’ energy of different frequencies on different places along the organ of Corti. Focusing magnifies stimulus intensity at each place for a small range of frequencies and attenuates all other frequencies. IHCs at each place then convey the intensity of vibration to their connected nerves. They don’t have any ability to separate frequencies so that task has to be completed before IHCs become involved or hearing quality will suffer (Figure 2).
But there is a problem. IHCs are insensitive mechano-receptors, having thresholds of around 60dBHL. Even worse, they only operate over a restricted intensity range of 40-50dB whereas our hearing requires more than 100dB range. The action of OHCs helps overcome both problems. The energy that OHCs add to the stimulus provides a large amount of amplification to weak stimuli but is insignificant in relation to strong sounds, which are not amplified. This ‘limitation’ of OHCs has the beneficial effect of compressing the range of vibrations into a much smaller range that IHCs can accommodate. When OHCs are damaged, hearing sensitivity, frequency selectivity and sound compression is lost. We fit hearing aids with amplification and compression – but cannot restore frequency selectivity.
OAEs are our only practical window on this vital cochlear process. Are we using them to maximum advantage? Research aimed at developing new clinical OAE applications is focusing on OHC’s role in the cochlea and on how and when OAEs are generated. When sound causes OHCs to ‘twitch’ some of their energy escapes back to the ear canal. Two processes are involved. When the highly magnified travelling basilar membrane wave hits tiny anatomical irregularities some of its energy is reflected back to the middle ear where it vibrates the ear drum and creates sound – a kind of ‘echo’. Separately the compressing action of OHCs creates distortions which are barely audible – but if two tones about a third octave apart are received then these distortions travel back to the ear drum transferring distortion to the ear canal sound (Figure 3).
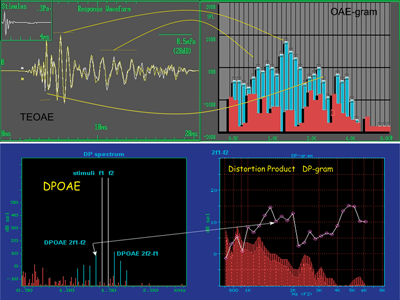
Figure 3. The two classic OAEs measurements, TEOAE and DPOAE, have changed little in 30 years. TEOAEs (top) are collected as a waveform (left) and then transformed into a TE-gram frequency spectrum display (right). DPOAEs (bottom) are collected as intermodulation distortion spectra (left) in response to two tones f1 and f2. The level of one of these distortions (2f1-f2) is plotted (right) as f1 and f2 are incremented, forming a DP-gram. Many other OAE measurements are possible and some could be even more useful.
These two processes account for the two commonly used OAEs tests. The echo or reflection emissions are typically observed using click sounds and are known as transient or click evoked OAEs (TEOAEs or CEOAEs). The compression or distortion product emissions are known as DPOAEs. Either form can be used for hearing screening. TEOAEs are fast and sensitive to as little as 30dB of hearing loss but capturing high frequencies is difficult. DPOAEs reach higher frequencies and are more ‘programmable’. With higher stimulus levels DPOAEs are recordable from ears with greater hearing loss (up to 60dB). But DPOAEs tell us different things depending on the stimulus levels and ratios used.
Both TE and DPOAEs give frequency specific information. Charts of OAE intensity across frequency can be compared to the audiogram – but ‘OAE-grams’ are definitely not to be thought of as equivalent to audiograms! Audiograms record ‘total hearing sensitivity’ – ‘ear to consciousness’, whereas OAE-grams only record the activity of OHCs which feed the IHCs, and even that is only true with normal middle ear function. Auditory brainstem responses (ABR) confirm the transfer of stimulation from OHC, to IHC, to auditory nerve.
“OAEs are a functionless leakage of OHC ‘twitch energy’, back to the ear canal.”
Comparison of OAE and ABR tests has great diagnostic value. Good OAEs and abnormal ABR indicates auditory neuropathy, but that’s not a single entity. Between OHCs and brainstem there is the micro-mechanical ‘pumping action’ of the organ of Corti, the IHC transduction mechanism, IHCs’ auditory nerve synapses, and the auditory nerves themselves. Pathology can occur in each process so today we consider OAE/ABR conflicts as indicative of an auditory neuropathy spectrum disorder including IHC dysfunction, and neural desynchrony. Reduction in the density of IHCs’ nerve connections, called synaptopathy, is being associated with ‘hidden hearing loss’ and may not be detectable by either OAEs or ABR.
It is clear that OAEs should be considered an essential part of the differential diagnostic test battery, not just a screening method. In the clinic today OAEs are often only used to confirm what is already suspected to be sensory hearing loss, rather than to gain additional information or to monitor cochlear condition over time. Most clinical OAE instruments are capable of more than screening, but even so their capabilities seriously lag behind developments in OAE recording methods, new knowledge about the cochlea and its pathologies. That was the unanimous conclusion of a high level NIDCD sponsored ‘OAE Workshop’ this February at the University of Southern California. The call was for a new generation of OAE instruments, and for ‘forward pressure level calibration’ (FPL) to be employed to provide more accurate stimulus levels at higher frequencies, helping to accommodate differing probe fittings and delivering the accuracy, range and reliability essential for early diagnosis and for ototoxic drug or long term cochlea monitoring (Figure 4).

Figure 4. DPOAEs carry rich information about the individual cochlea tested as seen in this DPOAE map. This information is not represented in today’s clinical DPgrams which are equivalent to just one line across the upper part of this map. DP maps record the intensity and phase of the DP 2f1-f2 for a wide range of frequencies and frequency ratios. They take longer to complete than a DP-gram but are a powerful way to detect change in the cochlea.
The workshop also emphasised that the incredibly rich information carried by OAEs when stimulated by complex and dynamic sound patterns is the key to better understanding of the cochlea and pathology types. But these measurements are not yet available for wider use in clinical research. The meeting heard of new discoveries and techniques – for example, that different aspects of OAEs (short vs long latency, reflection vs distortion, high vs low stimulus levels) relate differently to ageing and pathologies; that a hearing-loss-related mutation affecting the tectorial membrane, exaggerates spontaneous OAEs; that OAE mapping is helping pin down the origin of new emissions types and detect stressed regions in the cochlea; that cochlear compression can be gauged with OAEs.
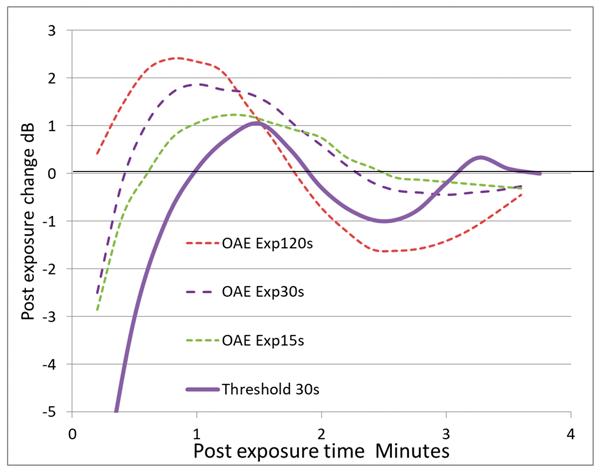
Figure 5. The action of the cochlea’s stabilising mechanism can be seen with OAEs. After being challenged by a 30s strong low frequency sound, hearing threshold at 1kHz (solid trace) recovers to normal in about 60s, but curiously then overshoots, becoming better than normal at 90s before showing another loss then stabilising. OAEs (dashed lines) after 15, 30 and 120s exposures show similar behaviours. This can be understood as a negative feedback system with a delay. It works to stabilise hearing sensitivity against stresses and is probably biochemical in nature.
Surprisingly it is still not known exactly how the cochlear amplifier is created by OHCs, or exactly how OAEs are generated or how the cochlea keeps itself stable at optimum performance. The cochlea’s homeostatic mechanisms are poorly understood but have the capacity to hide the onset of cochlea pathology from current audiological tests. OAEs are capable of revealing that process in action for example when OAEs slowly ‘bounce’ for two minutes after a loud sound (Figure 5). Ototherapeutics are on the horizon. Their effectiveness will almost certainly be monitorable with OAEs.
The next 10 years should see great developments in clinical OAE applications.





