Developing safe, reliable and efficient ways to deliver therapeutics into the cochlea is a key challenge. In this article, the authors share their work to develop microneedles to address this challenge.
The era of precision medicine of the inner ear is upon us [1,2]. With the identification of over 100 deafness genes and the recent demonstration of mammalian hair cell regeneration, potential of stem cell therapy, and the ease of gene editing technology, we are on the cusp of molecular therapy for inner ear disorders.
However, a significant impediment to implementing precision medicine for inner ear disorders is reliable access to the inner ear for diagnostic testing and therapeutic delivery while preserving hearing function and maintaining cochlear architecture. Protected by one of the hardest bones in body, the cochlea is nearly an impenetrable structure frustrating both bacteria and clinicians trying to gain access to it. As a result, a means for accessing the inner ear for diagnostic aspiration, as well as reliable delivery of agents into the inner ear for therapeutic purposes remains a formidable challenge. In the absence of precise diagnosis facilitated by sampling of inner ear fluid for electrochemical, RNA or proteomic analysis, precise intervention is not possible.
Intracochlear delivery
Options for intracochlear delivery include systemic administration, intratympanic injection or direct introduction into the cochlea. Systemic administration of the therapeutic agent has the drawback of systemic toxicity. Intratympanic (IT) delivery is hampered by variable efficacy and toxicity as it relies on the diffusion properties of the round window membrane (RWM), and injected medication can leak down the eustachian tube, be impeded by scarred round window niche, escape out of the external canal, or be sequestered in the middle ear. Placement of therapeutic agent directly adjacent to the RWM is inevitably limited by the unpredictable rate of molecular transport across the RWM. Importantly, aspiration of perilymph for diagnostic purposes is not possible with any of these methods.
"Based on this innovative work, we are on the threshold of launching the use of diagnostic and therapeutic microneedles in humans for facilitating precision medicine of the inner ear!"
Direct intracochlear drug administration has been shown to be superior to IT injection and results in significantly higher and less variable drug levels; in addition, there is a much smaller concentration gradient from base to apex, as is typical of IT injection, resulting in a more even distribution of material. Various methods of intracochlear delivery have been developed, including osmotic mini-pump infusion or microinjection into the scala tympani via the RWM, infusion or microinjection into the scala tympani through a cochleostomy, and injection into the endolymphatic sac accessed from the posterior cranial fossa. Recently, clinical trials have been proposed that combine cochlear injection with fenestration of the cochlea, stapes footplate or vestibular labyrinth to allow egress of fluid. All of these methods involve breaching the inner ear and consequently risk hearing impairment from traumatic disruption of the cochlea.
Accessing the inner ear through its only membranous structure – the three-layered round RWM that protects the inner ear from middle ear pathology – is an attractive portal for intracochlear access. Microneedle technology offers an elegant solution to overcome the difficulties of sampling cochlear fluid and facilitating reliable and predictable intracochlear delivery across the RWM without anatomic or functional damage [3]. To design needles that make controlled perforations while preventing undesired rips and tears in the RWM requires knowledge of its microanatomy and mechanical properties, as investigated in our laboratory [4].
Microneedles
Microneedle technology enables safe sampling of cochlear fluid and facilitates reliable and predictable intracochlear delivery across the RWM without anatomic or functional damage. Using microneedles, drug concentrations within the cochlea/semicircular canals can be controlled with a precision that IT injections simply cannot provide. The application of microneedles is not a departure from current clinical practices, but rather a natural progression from what is now in practice, in the setting of new technological advances. We believe microneedles – capable of direct aspiration of cochlear fluids for diagnostics followed by direct intracochlear injection for treatment across the RWM – to be a key enabling technology for precision medicine of the inner ear.
Microneedle design
Based on the mechanical properties of the RWM, we have designed, fabricated, and tested in vitro and in vivo in animal and human temporal bones, several types of microneedles. To overcome the limitations of other technologies to manufacture microneedles, we developed a unique set of nanomanufacturing methodologies to fabricate ultra-sharp microneedles from several different materials, including hard polymers and metallic microneedles, using a method we refer to as two-photon templated electrodeposition (2PTE) [5]. The microneedles can be hollow or solid, with a tip radius of curvature as sharp as 500nm and shaft diameters ranging from 20µm to 1mm [6]. The hollow microneedles have a 35µm diameter lumen through which fluids can be aspirated or injected at a rate of about 1 µL/min [3]. Figure 1 shows two such microneedles that are mounted onto blunt hollow stainless-steel needles for deployment.

Figure 1. Microneedles for perforation of round window membrane: A) Hard polymer microneedle mounted on 30 gauge hollow blunt stainless steel needle; B) Metallic microneedle mounted on 24 gauge hollow blunt stainless steel needle. Courtesy of authors.

Figure 2. a) Confocal microscopy image of guinea pig RWM perforated by microneedle with 100µm diameter; b) Higher magnification image near the perforation. The major axis of the lens-shaped perforation is aligned with the fibres of connective tissue, showing separation of the connective firers rather than scission. The small dark circular features are cellular debris. Image courtesy of Aksit A, et al [4] with permission.

Figure 3: a) Single 150mm microneedle perforation (white arrow) of human cadaveric round window membrane visualised using confocal microscopy. Collagen and elastin fibres appear continuous along the boundaries of the perforation, demonstrating separation not scission of connective fibres during perforation; b) Scanning electron microscopy image (SEM) of a representative microneedle after perforation, which shows minimal signs of blunting and bending at the tip, and is otherwise indistinguishable from unused microneedles. From Chiang H et al [6] with permission.
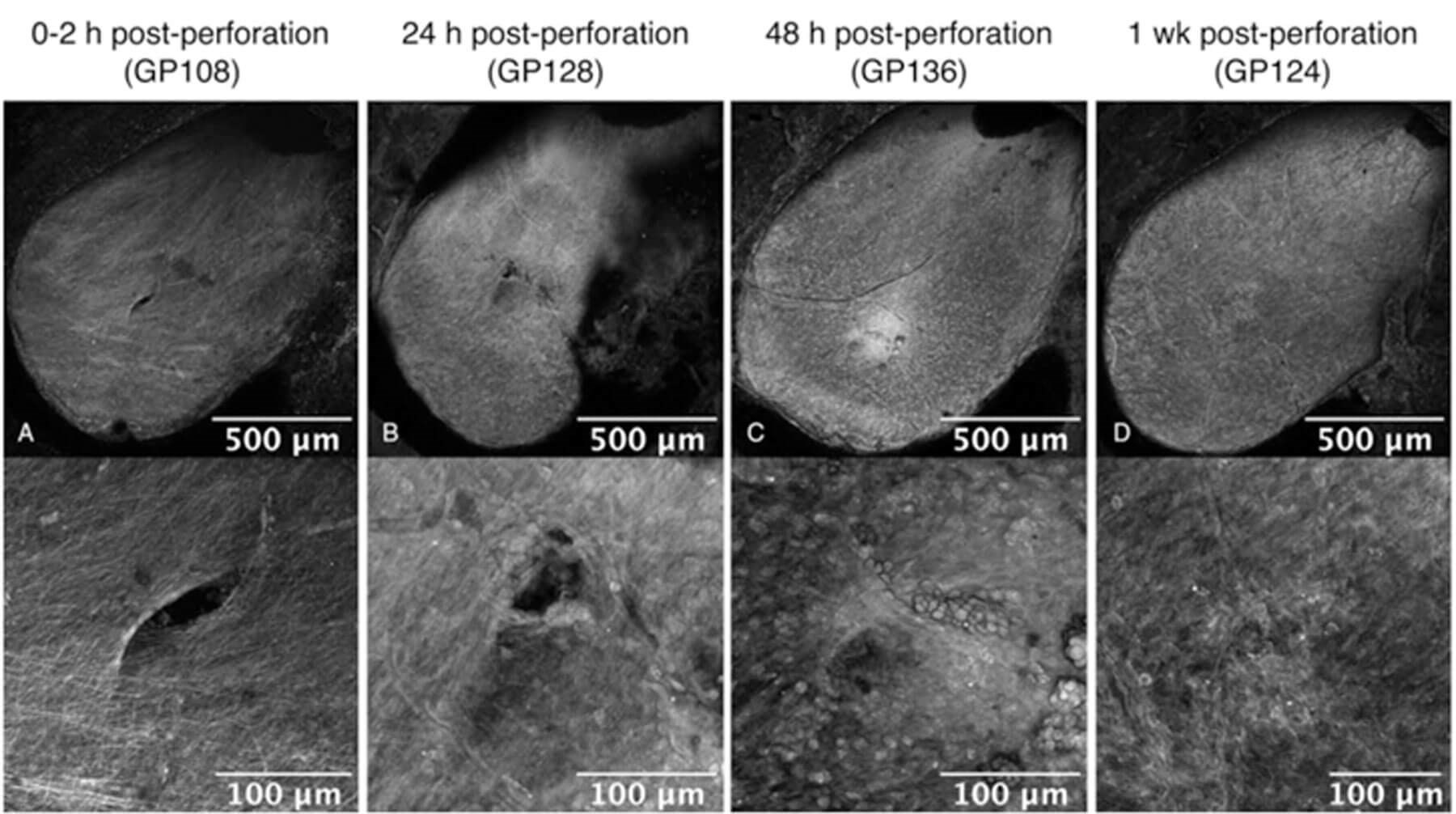
Figure 4. Guinea pig in vivo perforations of round window membrane healing over time: RWM perforations under x10 magnification (top) and x20 magnification (bottom) at (A) zero-to-two hours, (B) 24 hours, (C) 48 hours, and (D) one-week post-perforation, respectively. From Yu M et al [7] with permission.
Precision medicine of the inner ear
Using these microneedles, we have shown that a single microneedle or a microneedle array can create precise, accurate and reliable perforations across the RWM while predominantly separating, rather than cutting, the connective fibres in a guinea pig model (Figure 2) [4] and in human cadaveric RWM (Figure 3) [6]. Significantly, with this ‘separation not scission’ perforation mechanism, the RWM begins to heal after in vivo guinea pig RWM perforation by 24 hours, and is completely healed within 48-72 hours post-perforation (Figure 4) [7]. Importantly, there was no hearing loss associated with their use, as tested with compound action potential (CAP) and distortion product otoacoustic emissions (DPOAE) [7]. We have similarly used a hollow microneedle to aspirate 1µL of guinea pig perilymph without any damage for proteomic analysis identifying over 400 proteins demonstrating the utility of cochlear aspirate in diagnosis [3]. Injection with the same microneedle led to distribution of the fluorescent marker within the cochlea. A highly innovative dual lumen microneedle design will make possible delivery of significantly larger volume of therapeutic agent into the inner ear. Based on this innovative work, we are on the threshold of launching the use of diagnostic and therapeutic microneedles in humans for facilitating precision medicine of the inner ear!
References
1. Szeto B, Chiang H, Valentini C, et al. Inner ear delivery: Challenges and opportunities. Laryngoscope Investig Otolaryngol 2020;5(1):122‑31.
2. Valentini C, Szeto B, Kysar JW, Lalwani AK. Inner Ear Gene Delivery: Vectors and Routes. Hearing Balance Commun 2020;18(4):278-85.
3. Szeto B, Aksit A, Valentini C, et al. Novel 3D-printed hollow microneedles facilitate safe, reliable, and informative sampling of perilymph from guinea pigs. Hear Res 2021;400:108141.
4. Aksit A, Arteaga DN, Arriaga M, et al. In-vitro perforation of the round window membrane via direct 3-D printed microneedles. Biomedical Microdevices 2018;20(2):47.
5. Aksit A, Lalwani AK, Kysar JW, West AC. Simulation assisted design for microneedle manufacturing: Computational modeling of two-photon templated electrodeposition. J Manuf Process 2021;66:211-9.
6. Chiang H, Yu M, Aksit A, et al. 3D-Printed Microneedles Create Precise Perforations in Human Round Window Membrane in Situ. Otol Neurotol 2020;41(2):277-84.
7. Yu M, Arteaga DN, Aksit A, et al. Anatomical and Functional Consequences of Microneedle Perforation of Round Window Membrane. Otol Neurotol 2020;41(2):e280-7.
Declaration of competing interests: Anil K. Lalwani, MD, and Jeffrey W. Kysar, PhD. are two of the co-founders of Haystack Medical, Inc., that will commercialize microneedle technology to facilitate medical access to the inner ear.