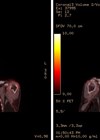
Conventional teaching tells us that hyposmia in chronic rhinosinusitis is due to mechanical obstruction of the olfactory cleft. But it might be that the story is slightly more complicated than that.
Olfactory dysfunction is a common feature of chronic rhinosinusitis (CRS) and yet, whilst it is a symptom rated highly by patients as a key complaint, it is often overlooked by clinicians. CRS is typically viewed as a conductive olfactory disorder, with the assumption being that occlusion of airflow to the olfactory clefts by nasal polyps is a mechanical issue that can be overcome through simple medical means or by surgical removal of polyps [1].
However, a closer look at this topic reveals that there is more to consider than meets the eye and that CRS may well represent a mixed conductive and sensorineural disorder in cases that present in secondary and tertiary care.
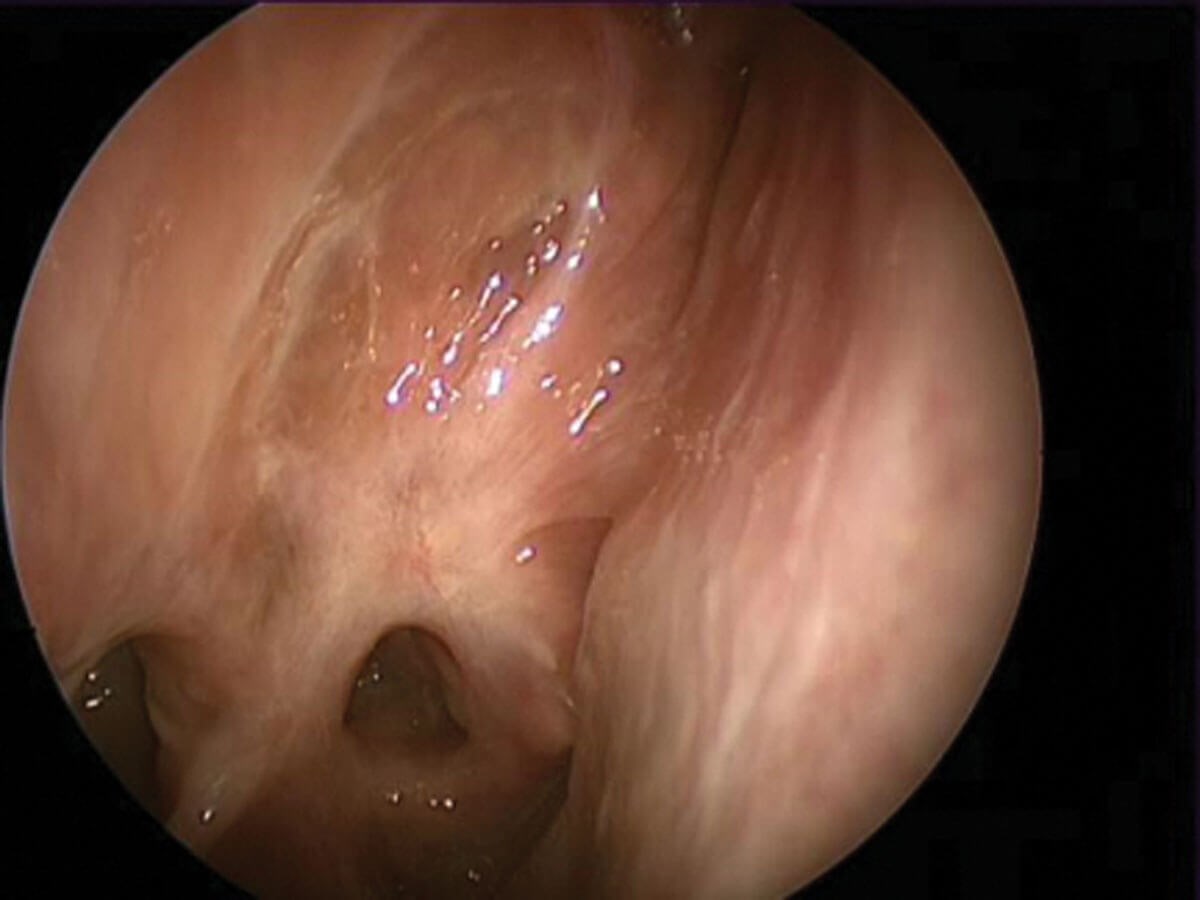
Scarred olfactory cleft following middle turbinate section.
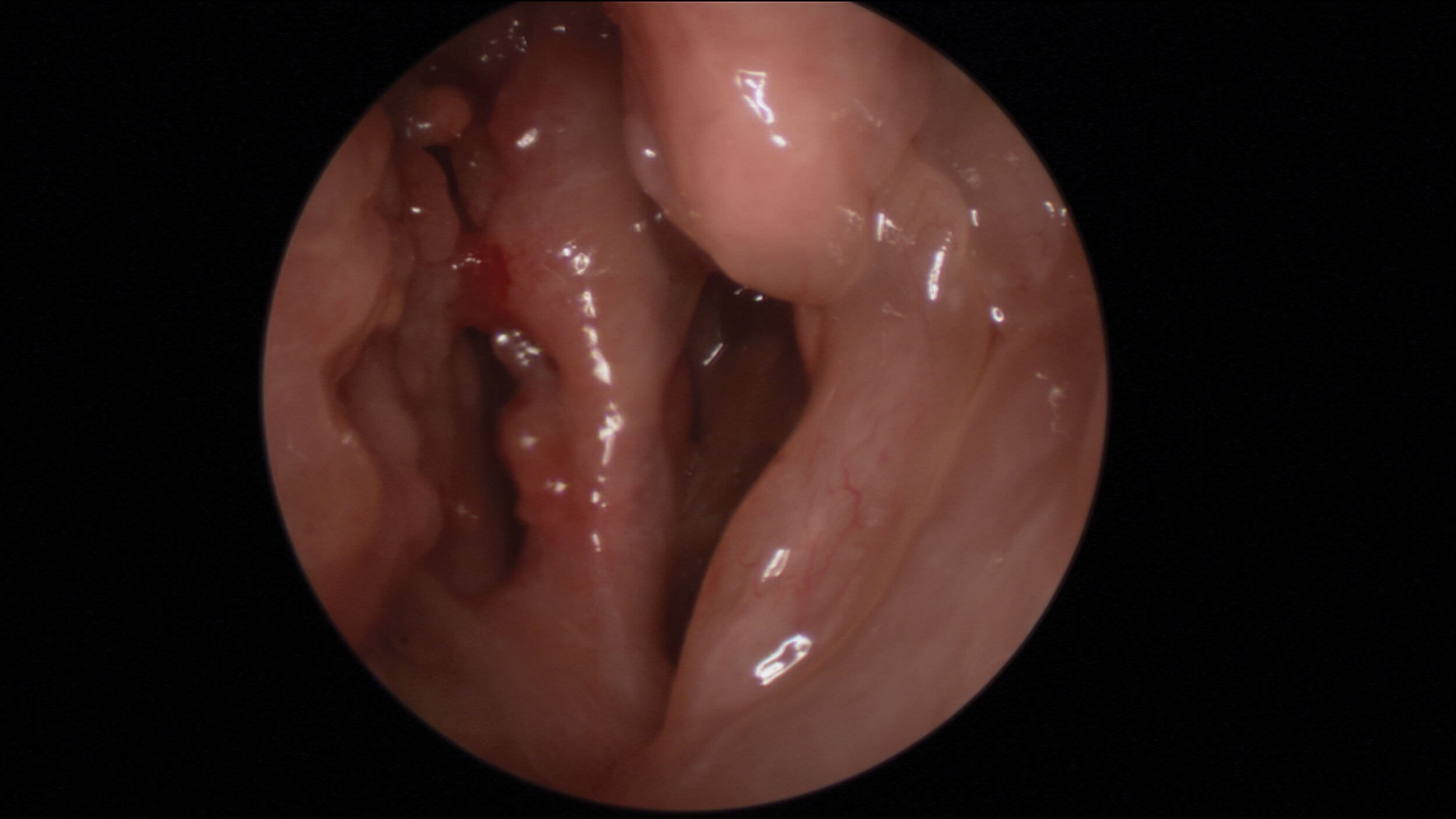
Oedematous olfactory cleft in NERD patient.
Mechanical obstruction
Supporting the notion that transmission to the OC due to mucosal oedema or polyps reduces quantitative olfaction is the positive response to oral and nasal steroids in many cases [1]. Furthermore, opacification of the olfactory cleft, as demonstrated on paranasal sinus CT imaging, has been shown to correlate well with olfactory function on psychophysical testing [2]. But it is not just oedematous mucosa that can occlude the olfactory clefts; thick eosinophilic mucin as seen in cases of non-steroidal exacerbated respiratory disease (NERD) and allergic fungal rhinosinusitis (AFRS) that can also accumulate in the olfactory clefts and spheno-ethmoidal recesses.
Neuroepithelial inflammation
An increasing body of evidence shows that there is a good correlation between eosinophilia and olfactory dysfunction [3]. Studies in both animal and human hosts suggest that inflammation in this specific cellular layer involves a temporary and reversible disruption of the process of olfactory binding and, thus, with quantitative olfaction [4].
"CRS may well represent a mixed conductive and sensorineural disorder in cases that present in secondary and tertiary care"
Respiratory metaplasia
If neuroepithelial inflammation persists, this may give rise to a more chronic inflammatory response whereby olfactory stem cell populations switch from regenerative to immune phenotypes [5]. The end process is one of neuroepithelial remodelling and eventual replacement with respiratory type epithelium which has no chemosensory functional capacity [6].
Upstream neurological impact
Olfactory bulbs are known for their plasticity in response to functionality of the olfactory system and, in the CRS, this is no different with evidence showing that olfactory bulb volumes are reduced in CRS patients [7]. It is therefore not surprising that the benefits of treatment for CRS on olfactory performance can be seen by the significant increase in olfactory bulb volume after treatment in patients with CRS [8]. But the story doesn’t stop at the olfactory bulbs, as further studies have shown reduced grey matter volume upstream of the olfactory bulbs in CRS patients, with evidence of both functional and structural plasticity in the olfactory cortex following surgical intervention for CRS [9].
Iatrogenic injuryIf
endoscopic sinus surgery is undertaken, it is essential that due care is taken with areas of the nasal cavity bearing olfactory epithelium, including the middle and superior turbinates and the upper nasal septum. More radical techniques that remove such areas not only have the potential to remove the cumulative olfactory neuroepithelial surface area but, in case of NERD and AFRS, it is not uncommon to see neo-turbinate formation from crops of nasal polyps that occlude the olfactory cleft. It is likely that more aggressive surgery goes hand in hand with more aggressive endotypes of CRS and that iatrogenic and neuroepithelial insults occur in tandem.
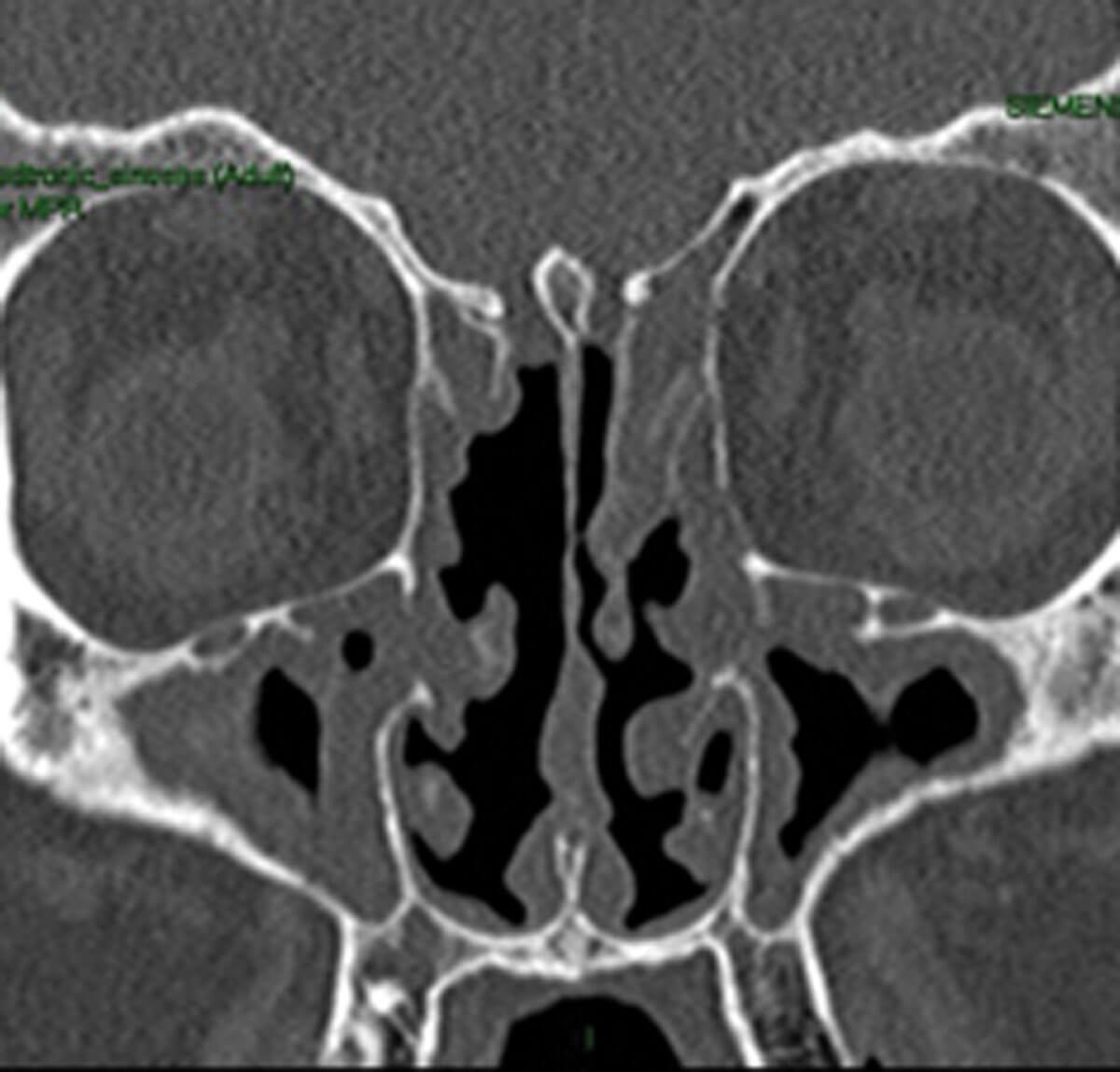
Radiological evidence of partial sinus surgery and middle turbinate removal.
Qualitative olfactory dysfunction
Again, with many of the assumptions around olfactory dysfunction in CRS, it is commonly thought that the overriding problem is one of quantitative dysfunction. However, patients with CRS do experience parosmia and phantosmia [10] but may also have a genuine reason to report ‘cacosmia’. Mucopurulent secretions and sinus mycelia can often have characteristic odours, and any rhinologist that takes the time to remove such secretions from the nose/sinuses can attest to the malodour of certain secretions. The clue here can be of close family members and friends reporting the malodour in addition to the patient, rather than an account of sensing an odour without any apparent stimulus.
Managing olfactory dysfunction
CRS patients clearly seek optimisation of the olfactory function from their treatment, so it is inherent that clinicians ensure the recommended treatment enables this as much as possible. Following clear guidelines on the management of CRS such as EPOS2020 [11] is a clear starting point. Oral corticosteroids will typically indicate the underlying potential for restoration of olfactory function and, if the response from short courses wanes, then the clinician will need to consider additional tools to attempt to maintain lasting benefits from treatment.
"It is likely that more aggressive surgery goes hand in hand with more aggressive endotypes of CRS and that iatrogenic and neuroepithelial insults occur in tandem"
Endoscopic sinus surgery may help to achieve better disease control, and meticulous primary surgery with preservation of olfactory epithelial surfaces is key to avoid iatrogenic injury. Postoperatively, steroid irrigations can be a useful starting point but use of special atomisers (e.g. laryngeal) can help to target the olfactory clefts specifically with topical steroids and help to achieve localised control that benefits olfactory performance. Finally, we are now seeing the advent of monoclonal antibodies in managing CRS, and the early trials have already shown how these agents beneficially impact on olfactory performance in the recipients [12]. As our understanding of how to better treat the sensorineural olfactory disorders progresses, it is likely that we will see further advances in treatment potential for this specific aspect of CRS management.
References
1. Alobid I, Benítez P, Cardelús S, et al. Oral plus nasal corticosteroids improve smell, nasal congestion, and inflammation in sino-nasal polyposis. Laryngoscope 2014;124(1):50-6.
2. Vandenhende-Szymanski C, Hochet B, Chevalier D, Mortuaireet G. Olfactory cleft opacity and CT score are predictive factors of smell recovery after surgery in nasal polyposis. Rhinology 2015;53(1):29-34.
3. Soler ZM, Sauer D, Mace J, Smith TL. Impact of mucosal eosinophilia and nasal polyposis on quality-of-life outcomes after sinus surgery. Otolaryngol Head Neck Surg 2010;142(1):64-71.
4. Pozharskaya T, Lane AP. Interferon gamma causes olfactory dysfunction without concomitant neuroepithelial damage. Int Forum Allergy Rhinol 2013;3(11):861-5.
5. Chen M, Reed RR, Lane AP. Chronic Inflammation Directs an Olfactory Stem Cell Functional Switch from Neuroregeneration to Immune Defense. Cell Stem Cell 2019;25(4):501-13.e5.
6. Jafek BW, Jafek BW, Murrow B, et al. Biopsies of human olfactory epithelium. Chem Senses 2002;27(7):623-8.
7. Rombaux P, Potier H, Bertrand B, et al. Olfactory bulb volume in patients with sinonasal disease. Am J Rhinol 2008;22(6):598-601.
8. Gudziol V, Buschhüter D, Abolmaali N, et al. Increasing olfactory bulb volume due to treatment of chronic rhinosinusitis-a longitudinal study. Brain 2009;132(Pt 11):3096-101.
9. Whitcroft KL, Noltus J, Andrews P, Hummel T. Sinonasal surgery alters brain structure and function: Neuroanatomical correlates of olfactory dysfunction. J Neurosci Res 2021;99(9):2156-71.
10. Philpott C, Dixon J, Boak D. Qualitative Olfactory Disorders: Patient Experiences and Self-Management. Allergy Rhinol (Providence) 2021;12:21526567211004251.
11. Fokkens WJ, Lund VJ, Hopkins C. European Position Paper on Rhinosinusitis and Nasal Polyps 2020. Rhinology 2020;58(Suppl S29):1-464.
12. Chong LY, Piromchai P, Sharp S, et al. Biologics for chronic rhinosinusitis. Cochrane Database Syst Rev 2021;3:CD013513.